Though people have studied and been fascinated by electricity and magnetism, including such luminaries as Benjamin Franklin, we can really trace the beginning of modern electromagnetic theory to one specific experiment in 1785, in which the French physicist Charles-Augustin de Coulomb quantitatively measured the force between two electrically charged objects. The law of attraction he found, known as Coulomb’s law, set the stage for all of our understanding and progress in electromagnetism, including the computer and internet you’re reading this on.
I was a little bored this week and it so happens I have an electronic copy of Coulomb’s original paper1 of 1785 and decided to translate it from French and write about it! As always, I used a mixture of my own crude understanding of French combined with Google translate to do this. Coulomb’s experiment is truly amazing, important and beautiful and it was quite fun to see the details of how he did it.
So who is Charles-Augustin de Coulomb? He was born in 1736 in Angoulême, France, to a wealthy family, and had education in science and mathematics in both Paris and Montpelier. He entered the École royale du génie de Mézières, a military engineering school, in 1760 and graduated in 1761. From there, he joined the French army and spent the next two decades doing engineering work including fortifications and soil mechanics. In the 1770s, he started writing papers on applied mechanics and in 1779 he wrote an important treatise on frictional forces, Théorie des machines simples, en ayant regard au frottement de leurs parties et à la roideur des cordages. It appears that his work on electrostatics was an offshoot of his work in studying friction.

Let us turn to his paper, which is titled “First Memoire on Electricity and Magnetism.” He would write seven memoires on the subject in total, though the first captures the most important discovery he would make.
In what amounts to the abstract of the paper, Coulomb writes:
Construction and use of an electric balance, based on the property that metal wires have of having a torsional reaction force proportional to the angle of torsion
Experimental determination of the law according to which the elements of electric bodies of the same kind of electricity repel each other.
The first sentence describes the technique, and the second the result; we will discuss each in more detail in what follows.
The paper properly begins with:
In a Memoir given to the Academy in 1784, I determined, based on experiment, the laws of the torsional force of a metal wire, and I found that this force was, in terms of the composition of the angle of torsion, the fourth power of the diameter of the suspension wire and the inverse of its length, by multiplying the whole by a constant coefficient which depends on the nature of the metal, and which is easy to determine by experiment.
Coulomb’s paper of the previous year2 was a mechanical study of the very small forces that apply when a hanging metal wire is twisted. The force is approximately proportional to the angle that the wire is twisted and the constant of proportionality depends on the diameter of the wire, its length, and the type of metal used.
In the same Memoir, I demonstrated that, by means of this torsional force, it was possible to measure with precision forces of very slight magnitude—such as, for example, one ten-thousandth of a grain. In that same Memoir, I presented a first application of this theory by seeking to evaluate the constant force attributed to adhesion in the formula expressing the friction of the surface of a solid body moving through a fluid.
The torsional force is very weak, which means that with the right experimental setup a torsional balance can be used to measure extremely small forces that would otherwise be undetectable. Coulomb notes that he already used such an approach to measure frictional forces on a solid body moving through a fluid. But he immediately recognized that it could be used for much more fundamental studies!
Today, I place before the eyes of the Academy an electric balance constructed upon the same principles; it measures with the utmost precision the electrical state and force of a body, however faint the degree of electricity may be.
I haven’t tried to describe a torsional balance in detail yet, because I wanted to wait and just show you Coulomb’s own beautiful drawing of his apparatus.
Although experience has taught me that, in order to conveniently perform several electrical experiments, it is necessary to correct some defects in the first balance of this kind that I had made, however, as it is the only one I have used so far, I will now give its description, noting that its shape and size can and should be varied according to the nature of the experiments one intends to perform. The first figure represents this balance in perspective, and here is the detail.
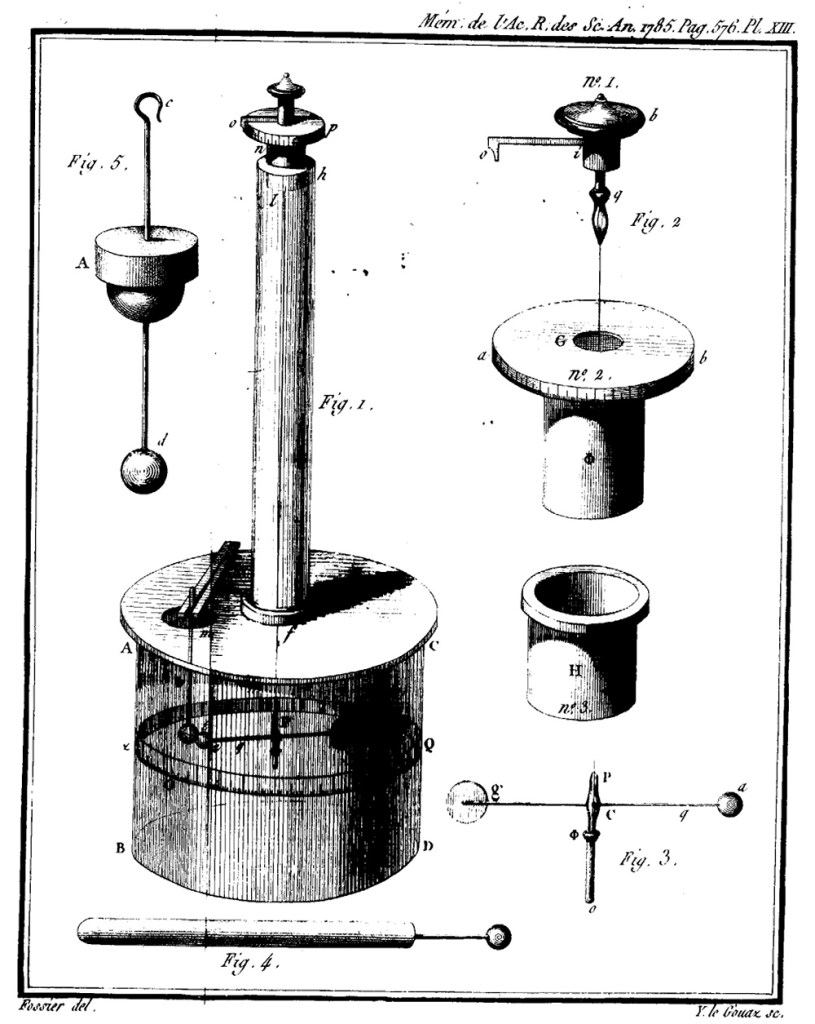
Figure 1 is the most important part here, so let’s describe the device in simple terms before letting Coulomb talk about it in more detail. We have a thin wire hanging down through the narrow cylinder into the large one. At the bottom of the wire is a cross-rod that is the actual balance. One side of this rod has a ball that can be electrified, and the other side just provides a counterweight. A second electrified ball can be dropped through an opening near the first one, and the angular deflection of the balance gives a measure of the force between the balls; this can be compared to the distance the balls are separated which gives an overall measure of how the force depends on distance between electrified objects.
For completeness, though, let’s go through Coulomb’s whole description! I break up some of his astonishingly long paragraphs for readability.
On a glass cylinder ABCD, 12 inches in diameter and 12 inches high, a glass plate of 1/2 inch diameter is placed, covering the entire glass cylinder; this plate is pierced with two holes approximately 20 lines in diameter, one in the middle, at f, above which rises a glass tube 24 inches high; this tube is cemented over hole f with the cement used in electrical apparatus; at the upper end of the tube at h is placed a micrometer of rotation, which can be seen in detail in the figure.
A “line” in this case is a ligne, a historical unit of length, eventually defined by convention as 443.296 lignes per meter. This unit was common in Coulomb’s time and even survives today and is in use by French and Swiss watchmakers! There are twelve lignes to one French inch of the era.
The micrometer at the top is an important piece; this allowed Coulomb to set a “zero” position for the micrometer. He describes in in more detail:
The upper part, no. 1, bears the button b, the index io, and the suspension clip q; This piece fits into hole G of piece no. 2. This piece, no. 2, is formed of a circle ab divided on its field into 360 degrees, and of a copper tube Φ which fits into the tube H, no. 3, welded inside the upper end of the tube or of the glass rod fh of figure 1.
The button is the foundation of the micrometer, the index indicates the orientation of the button, and the clip q holds the wire in place. Piece number 2 is marked into 360 degrees to allow quantitative orientation of the device.
The clip q, figure 2, no. 1, has approximately the shape of the end of a solid pencil holder, which can be tightened by means of the ring q; it is in the clip of this pencil holder, that the end of a very fine silver wire is grasped; the other end of the silver wire is grasped (fig. 3) at P, by the clip of a cylinder P of copper or iron, whose diameter is scarcely more than a line, and whose end P is bent, and forms a clip which is tightened by means of the sliding Φ. This small cylinder is hollow and pierced at C, to allow the needle ag (Fig. 1) to slide through it; the weight of this small cylinder must be substantial enough to keep the silver thread taut without breaking it. The needle—shown at ag in Fig. 1, and suspended horizontally at approximately half the height of the large vessel enclosing it—is formed either of a silk thread coated with sealing wax, or of a straw likewise coated with sealing wax, and terminates—along a length of 18 lines, from q to a —in a cylindrical rod of shellac. At the extremity of this needle is a small pith ball (made of elder pith) measuring two to three lines in diameter; at g is a small vertical vane of paper treated with turpentine, which serves as a counterweight to the ball at a and dampens the oscillations.
This lengthy paragraph gives the details of the clip supporting the silver wire at each end. The balance at the bottom is made of an extremely thin material, either a piece of straw or an actual silk threat coated with sealing wax. At a is the pith ball that will carry an electric charge. at g it is interesting to note that the counterbalance is a small slip of paper that will catch air and dampen any incidental oscillations of the balance that would make the measurement more difficult.
We said that the lid AC was pierced with a second hole at m; it is into this second hole that one inserts a small cylinder m Φ t, the lower part of which Φ t is made of shellac, and which is topped by a small ball, also made of elderwood; around the vase, at the height of the needle, one describes a circle τ Q divided into 360 degrees. For more simplicity, I use a strip of paper divided into 360 degrees, which I stick around the vase, at the height of the needle.
“Elderwood” here presumably refers to a ball made of the wood of the elderberry plant. This is the second ball that will be electrified, though it stays in a fixed position. To measure how much the balance twists due to the electric force, Coulomb stuck a strip of paper marked with degrees around the cylinder.
Next Coulomb describes the calibration of the device! Though I’ve known the basics of Coulomb’s experiment for some time, I’d never really looked into how he operated it in detail, so I found this particularly interesting.
To begin operating with this instrument, I proceed as follows: by positioning the lid, I align hole m approximately with the first division—or point o—of the circle τ o Q inscribed on the vessel. I set the index oi of the micrometer to point o (or the first division) of that micrometer. I then rotate the entire micrometer within the vertical tube fh until—when sighting along the vertical thread suspending the needle and through the center of the ball—the needle ag is seen to align with the first division of the circle τ o Q. Next, I insert the other ball—suspended by the thread m Φ t —through hole m, positioning it so that it touches ball a, and such that—when sighting along the center of the suspension thread and through ball t—one aligns with the first division (o) of the circle τ o Q. The balance is now ready to be employed for all operations; as an example, we shall now describe the method we utilized to determine the fundamental law governing the mutual repulsion of electrified bodies.
In short: he rotates the top glass plate so that hole m is aligned with the zero of the big cylinder. Then he sets the micrometer to its zero position, and rotates the whole thing until the two zeros align and the needle is aligned with the zero of the big cylinder. Then he puts the second ball through hole m, where it should push ball a aside and be aligned with the zero. Now the balls are touching and both the zero position of the balance and the zero position of the fixed ball are in agreement.
At last, we come to the key experiment, under a section “Fundamental law of electricity.”
A small conductor, fig. 4, which is nothing more than a large-headed pin, is electrified. This pin is insulated by inserting its point into the end of a stick of Spanish wax. The pin is then inserted into the hole m and made to touch ball t, which is in contact with ball a. Upon removing the pin, both balls are electrified with the same type of electricity and are driven apart at a distance that is measured by observing the corresponding division of the rotating circle through the suspension wire and the center of ball a. Then, the micrometer index is moved in the direction pno. The suspension wire is twisted, producing a force proportional to the angle of twist, which tends to bring ball a closer to ball t. By this means, we observe the difference to which different angles of rotation bring ball a towards ball t: by comparing the rotation forces with the corresponding differences of the two balls, we determine the law of repulsion.
The cleverness of the starting point is using a single electrified pin, illustrated again below, to electrify both balls simultaneously while they are touching. The two balls will end up with the same amount of electricity upon them, eliminating any possibility that variations in the two magnitudes have any effect. Ball a will be repelled from ball t, and will settle at a position where the torsion force of the wire balances the electrical force between the balls. Then, by turning the micrometer, Coulomb was able to adjust the relative orientation of ball a and could tally a list of the distance between the balls versus force between them.

Coulomb then describes some basic results. He gives very few numbers, though he presumably did many more experiments; evidently he just wanted to highlight the key observations.
I will only present here a few effects which are easy to repeat, and which will immediately demonstrate the law of repulsion.
First attempt. Electrified the two balls with the pinhead, the micrometer index responding to 0, the ball a of the needle moved away from the ball t by 36 degrees.
Second attempt. Having twisted the thread of the pendulum, by means of the o button of the micrometer to 126 degrees, the two balls came closer together and stopped at 18 degrees apart from each other.
Third attempt. Having twisted the suspension wire by 567 degrees by 567 degrees, the two balls came together at 8 and a half degrees.
These are all from the same electrification. The balls were electrified, and from the starting position, Coulomb found the balls were separated by 36 degrees. Then he twisted the micrometer to 126 degrees, in principle moving the two balls closer, and found that they got within 18 degrees of each other. Then he twisted the micrometer even further, by a total of 567 degrees — going over a full rotation — and found that the two balls were still repulsed at 8.5 degrees. Basically, the more he twisted the micrometer, the more torsional force he applied to force the balls together.
But what does it mean? Next we come to “Explanation and results of this experiment.” Coulomb writes:
When the balls are not yet electrified, they touch, and the center of the ball, suspended from the needle, is no more than half the diameter of two balls away from the point where the torsion of the suspension wire is zero. It must be noted that the silver wire lp, which formed the suspension, was 28 inches long and this wire was so fine that a foot of its length was only a few grains. By calculating the force required to twist this wire, acting at point a, four inches away from the wire lp or the center of torsion, I found, by the formulas explained in a Memoir on the laws of the force of torsion of metal wires, printed in the volume of the Academy for 1784, that to twist this wire by 360 degrees, it is necessary to employ at point a, acting with the lever an, four inches long, only a force of 1/340 of a grain, thus, as the forces of torsion are, as is proven in this Memoir, like the angles of torsion, the slightest force exerted between the two wires, forcefully moved them apart from each other.
In short, Coulomb was able to use his knowledge of the torsional forces that he determined in his previous paper to determine the quantitative force between the two electrified balls.
Here is the point where we should pay attention, because Coulomb with these two paragraphs initiated a new era of quantifying electromagnetic forces! He writes:
In our first experiment, where the micrometer index is at point o, we find that the balls are separated by 36 degrees, which simultaneously produces a torque of 36d = 1/3400 of a grain; in the second trial, the distance between the balls is 18 degrees, but since the micrometer has been twisted by 126 degrees, it follows that at a distance of 18 degrees, the repulsive force was 144 degrees: thus, at half the first distance, the repulsion of the balls is quadrupled.
144 degrees is exactly 4 times 36 degrees, which indicates that the force at 18 degrees separation is 4 times the force at 36 degrees separation!
In the third test, the suspension wire was twisted by 567 degrees, and the two balls were now only 8.5 degrees apart. The total twist, consequently, was 576 degrees, four times that of the second test, and it only took half a degree for the distance between the two balls in this third test to be reduced to half that of the second. It therefore follows from these three tests that the force exerted by two balls electrified with the same type of electricity on each other is inversely proportional to the square of the distances.
Emphasis mine in the final sentence! Here we have the famous inverse square law of Coulomb: the force between two electrified points is inversely proportional to the square of the distance between them.
Coulomb would build upon this result in his later memoires, showing that the law of attraction between opposing charges also satisfies an inverse square law, and that the force is proportional to the product of the charge magnitudes. But this precise measurement of an inverse square law was the real foundational discovery.
Coulomb was not quite done at this point, and added some “remarks” about his work.
By repeating the preceding experiment, we will observe that, by using a silver thread, however fine than the one we have used, which gives, for the force of torsion, at an angle of 5 degrees, only one thousandth of a grain, approximately, however calm the air may be, and however many precautions one takes, one cannot guarantee the natural position of the needle, or the torsion is zero, except to within 2 or 3 degrees. Thus, to have a first test to compare with the following ones, it is necessary, after electrifying the two balls, to twist the pendulum thread by 30 to 40 degrees, which, combined with the difference between the two balls observed, will give a sufficiently strong torsional force, so that the 2 or 3 degrees of uncertainty in the first position of the needle, when the torsion is zero, do not produce a significant error in the results. It should also be noted that the silver wire, which I used in this experiment, is so fine that it breaks at the slightest movement. I subsequently found it more convenient to use in experiments a suspension wire of almost double the diameter, although its torsional flexibility was fourteen to fifteen times less than that of the first. Before using this silver wire, care must be taken to hold it taut for two or three days under a weight approximately half that which it can bear without breaking. It must also be warned that when using this silver wire, it should never be twisted beyond 300 degrees, because beyond this angle of twist, it begins to stiffen and, as we have proven in the aforementioned Memoir, printed in 1784, reacts only with a force less than the angle of twist.
This comment is really about some practical details in the experiment. He notes that one gets inaccurate results with the micrometer in the neutral position, because the forces involved are so weak that the balance is easily perturbed. He also notes that a thicker wire works better for the experiment, because the thin wire can break easily.
The electricity of the two balls decreases slightly during the experiment; I have found that, on the day I performed the preceding tests, the electrified balls, being 30 degrees apart due to their repulsion, under a torsion angle of 50 degrees, are brought closer together by one degree in three minutes; but as I only used two minutes to perform the three preceding tests, one can, in these experiments, neglect the error resulting from the loss of electricity. If one desires greater precision, or when the air is humid, and the electricity is lost rapidly, one must, by a first observation, determine the rate or decrease of the electric current of the two balls in each minute, and then use this first observation to correct the results of the experiments that one wishes to carry out that day.
Here, Coulomb warns of something that should be familiar to everyone: static charges slowly dissipate with time. Within a few minutes the charge is significantly lost, but Coulomb notes that he did his experiments fast enough that this effect should not change the results.
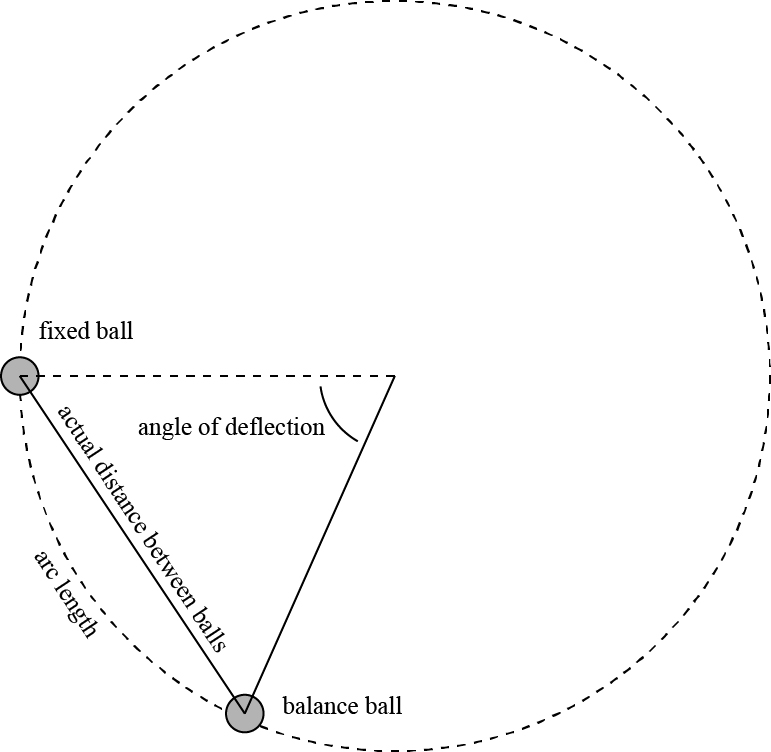
The distance between two balls, when they are separated from each other by their mutual repulsive force, is not precisely measured by the angle they form, but by the chord of the bow joining their centers; just as the lever at whose end the action is exerted is not measured by half the length of the needle, or by the radius, but by the cosine of half the angle formed by the distance between the two balls; these two quantities, one of which is smaller than the arc, and consequently reduces the distance measured by this arc, in the same way that the other reduces the lever, compensate each other in some way; And in experiments of the kind we are dealing with, one can, without significant error, stick to the evaluation we have given; if the distance between the two balls does not exceed 25 to 30 degrees, in other cases, it must be calculated rigorously.
Here Coulomb is simply noting a little geometry. Since the ball is fixed to rotate on a circular path, the actual distance between the balls is shorter than the arc length between the two balls, as illustrated below.
This also effects the measurement of the forces! The torsional force is directed along the arc of the circle, while the electrical force between the balls is directed along the actual distance between them. Coulomb notes that as long as the actual distance between the balls is less than 25 or 30 degrees, these differences will not cause significant errors in the measurement. (In the picture above, I used a roughly 60 degree angle to highlight the differences.)
As experience proves, in a well-sealed chamber, one can determine, with the first silver wire, to within 2 or 3 degrees, the position of the needle when the twist is zero, which gives, according to the calculation of the torsional forces proportional to the angle of torsion, a force of at most one forty-thousandth of a grain, the weakest degrees of electricity will be easily measured with this balance. For this operation, one passes, Fig. 5, through a Spanish wax stopper, a small copper wire c d, ending at c with a hook, and at d, with a small gilded elderberry ball, and one places the stopper A in the hole m of the balance fig. 1, in such a way that the center of ball d, seen by the suspension wire, corresponds to point o of the circle τ o Q by approaching an electric body to the hook c, however weak the electricity of this body, the ball separating from ball d gives signs of the electricity, and the distance between the two balls measures the force, according to the principle of the inverse ratio of the square of the differences.
Here Coulomb modifies his experiment a little, replacing the fixed ball by a ball with a hook protruding from the device, as shown below (turned sideways).
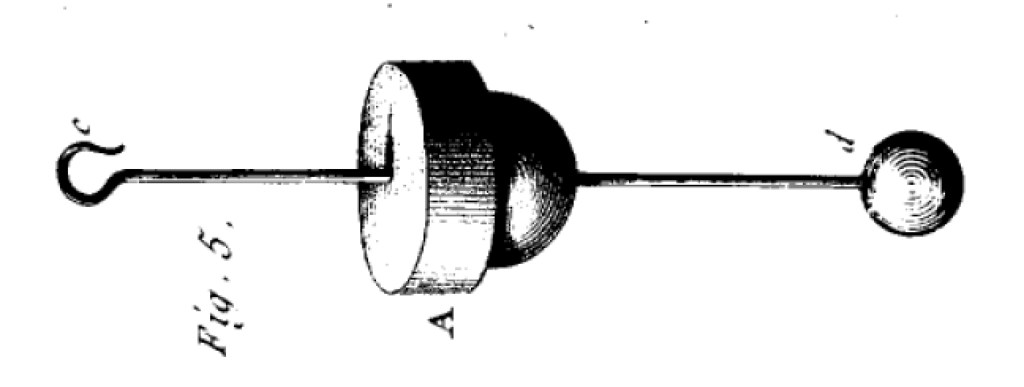
The idea is using the device to measure the strength of electric charge rather than the law of electric forces! Now that the force of electricity is known to satisfy an inverse square law, Coulomb suggests that one can bring an electrified object near the hook of Fig. 5. This will induce a proportional amount of electricity in the ball d, and the amount of electricity can be quantified by how much the balance arm deflects. Coulomb thus invented the first quantitative detector of electric charge.
Coulomb concludes with some extra cautionary details, which I include for completeness.
But I must warn that, since these first experiments, I have made different small electrometers, according to the same principles of torsional force, using for the suspension wire, a silk thread, such as is strong from the cocoon, or an Angora goat hair. One of these electrometers, which has almost the same shape as the electric balance described in this Memoir, is much smaller; it is only 5 to 6 inches in diameter, with a one-inch stem; the needle is a small shellac thread 12 lines long, ending in a very light, small circle of tinsel. The needle and the tinsel peel off about a quarter of a grain; the suspension thread, as it comes from the cocoon, being 4 inches long, has such flexibility that, by acting with a lever arm of one inch, only one sixty-thousandth of a grain is needed to twist it a full circle or 360 degrees: by presenting in this electrometer to hook C of Fig. 5, an ordinary stick of Spanish wax electrified by friction 3 feet from this hook, the needle is driven out to more than 10 degrees. We will describe this electrometer in detail in the following section, when we want to determine the nature & the degree of electricity of different bodies, which, by rubbing against each other, acquire a very high degree of electricity.
Coulomb thus ends his paper noting that he has made even more precise electrometers that can detect frictional electric charges even when they are brought three feet from the hook of Figure 5! He gives a hint of what comes in his second memoire: looking at the degree of electricity of different bodies using his electrometers. But for now, we have seen enough: we have seen how Coulomb measured, for the first time, the law governing the force between electric charges. From this foundation would be built all of modern electromagnetic theory and all of the benefits that have come from it.
***********************************
- Coulomb (1784) “Recherches théoriques et expérimentales sur la force de torsion et sur l’élasticité des fils de metal,” Histoire de l’Académie Royale des Sciences, pages 229–269.
- Coulomb (1785) “Premier mémoire sur l’électricité et le magnétisme”, Histoire de l’Académie Royale des Sciences, pages 569–577.