One of the joys of physics, and science in general, is that even seemingly mundane objects occasionally yield physical surprises. A great example of this made the news about a month ago: the observation that, under the right circumstances, x-rays can be generated by the peeling of Scotch tape! The phenomenon is an extreme example of the phenomenon of triboluminescence, and I thought I would take a closer look at the research results, which appeared in Nature.
First, a quick but important notice: THERE’S NO REASON TO WORRY ABOUT USING STICKY TAPE AT HOME! As we will note below, the x-ray effect is only significant when tape is peeled in a high vacuum. Such a condition obviously does not occur without special preparation. So the wrapping of Christmas packages can continue without fear.
It’s worth taking a moment to explain why this seems like such a surprising result in the first place. Interaction energies in normal chemical interactions tend to be no greater than 10’s of electron volts; for instance, it takes 13.6 eV to ionize a hydrogen atom. If the reaction releases a photon, this puts the wavelength of the photon at best in the ultraviolet or visible range, with an energy several orders of magnitude lower than the keV or MeV of x-rays. X-ray emission from atoms under normal circumstances comes only from nuclear processes, e.g. the decay of an atomic nucleus. Chemical reactions seemingly don’t have enough ‘oomph!’ to generate x-rays.
Electromagnetic radiation can be generated by any acceleration of electric charges, however, and with the right system this can readily produce x-rays. X-rays were in fact discovered by Wilhelm Röntgen in 1895 while he was experimenting with cathode ray tubes (which is, in essence, a device which accelerates electrons along a vacuum tube). Electrons, traveling from a cathode to an anode, are decelerated sharply on interaction with the anode. An illustration of such a tube is shown in a Wikipedia picture below, with the cathode on the left:

This process of ‘braking’ releases radiation, known as Bremsstrahlung radiation. This radiation will include x-rays if the ‘braking’ is strong enough, i.e. if the electrons were moving fast enough to begin with.
Another example of x-ray emission from acceleration is the phenomena of synchrotron radiation, in which particles in high-energy accelerators shed x-rays as they circulate. Initially, such radiation was seen as a hindrance to performing high-energy physics experiments, but now it is used at places such as Argonne National Laboratory as a source of x-rays for other experiments.
These examples involve the acceleration of charged particles to high velocities. There seems at first glance to be no such process going on when one peels a roll of tape, which is what makes the result of the University of California research group so interesting.
The phenomena is an example of a process known as triboluminescence, which in short refers to the release of light from crystalline structures when they are rubbed, scratched or broken. The phenomenon has been well-known for quite some time, dating back to the early 1600s; Jennifer at Cocktail Party Physics has written a nice description of the history of triboluminescence and some of the current developments (and scooped me again!). A classic example (which is celebrating its 90th anniversary), is the ‘spark’ produced when biting down on a Wint-O-Green Life Savers (for your sanity, stop the video after the first 11 seconds):
Interestingly, the most sensational parts of the Nature paper are not new; in fact, they are quite old! As the authors point out, triboluminescence from Scotch tape was observed back in 1939*, and evidence that x-rays can appear in triboluminescence was suggested in 1930** and confirmed in 1953***.
The first hint of x-rays in triboluminescence appeared in a paper by J.W. Obreimoff on, “The splitting strength of mica.” In addition to studying the ability of polished glass plates to readhere to one another after splitting, Obreimoff describes some electrical phenomena which appear when the mica is split in a high vacuum. In anticipation of the results relating to Scotch tape, Obreimoff notes,
If split in darkness, mica becomes slightly luminescent (triboluminescense). This is due to electric discharges between the mica surfaces through the air. If we split them under an air pressure of 1.0-0.1 mm. mercury the glow spreads to all the air in the vessel and is similar to the glow of a Geissler tube. In a high vacuum (
mm. mercury) the glass of the vessel fluoresces like an X-ray bulb. The light is feeble and can be observed only after the eye has rested about 3 minutes in darkness.
This fluorescing of the glass of the vessel suggested the presence of x-rays, and it only appears when a vacuum is present, just like the tape experiments to be described.
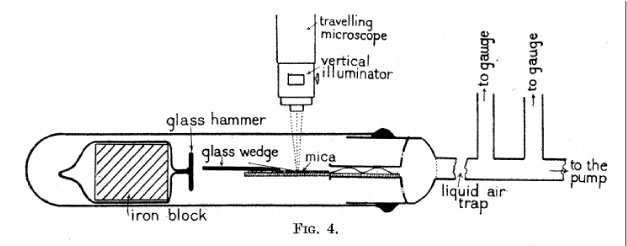
I can’t resist pointing out how one actually splits a piece of mica which is isolated in a high vacuum! I take the liberty of reproducing the figure of the experimental apparatus from the paper:
A glass wedge is placed under the strip of mica being separated, and a glass hammer, attached to a glass-encased iron block, is adjacent to it. An electromagnet is used to manipulate the block from the outside and smack it against the glass wedge.
In 1939, N.E. Harvey wrote a ‘discussion’ of the luminescence of adhesive tape. Harvey gives a rough explanation of the process of luminescence:
It is apparent that these phenomena have a decidedly electrical flavor. A sheet of collodion stripped from glass or ebonite has a high negative charge, leaving the glass and ebonite positive. It is attracted to the glass with considerable force and sticks to the hand and other objects. The sign of the charge is easily determined by pith ball experiments.
The explanation of such luminescence appears to be this: whenever two surfaces are separated from each other the capacity diminishes and the voltage rises until a discharge takes place, exciting the surrounding gas to luminesce. It is not possible to prove that mica sheets or tire tape, surgeons’ tape or Scotch tape are oppositely charged as a whole when pulled apart, but there are no doubt local positive and negative regions developed, the discharge between them giving rise to luminescence.
That a discharge does actually take place can be readily shown by stripping surgeons’ tape or Scotch tape in an atmosphere of 2 to 4 cm Hg pressure of Neon gas. Then the luminescence is reddish instead of yellowish.
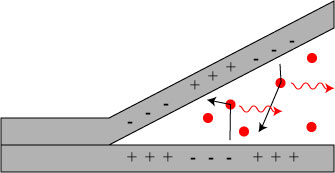
A rough diagram of the hypothesized process is shown below:
As the tape (or mica) is separated, local regions of separated + and – charge are created. When these regions are separated far enough, the electric force pulls the electrons from the negative side to the positive side. Along the way, though they collide with (and excite) gas molecules, which then produce fluorescence. The color of fluorescence depends on the type of gas molecules present.
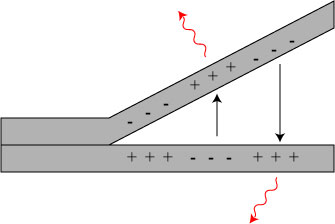
How can we get x-rays from such a process? If we evacuate the chamber in which the separation is taking place, the electrons are now free to travel the entire distance from one surface to another. With a large enough potential difference between the surfaces, they act essentially as a natural cathode ray tube! Electrons travel from the cathode (- surface) to the anode (+ surface), and when they arrive they are slowed down by Bremsstrahlung radiation:
This is the sequence of events suggested by the authors of the recent Nature article. But, as we’ve noted, triboluminescence has been observed from adhesive tape before, and x-ray emission from triboluminescence has been observed; so what is new about the current crop of experiments?
There are several new aspects. First, no modern studies of x-ray emission in the peeling of Scotch tape had been performed, and it is striking that such an inexpensive and mundane material could have such properties. Second, they demonstrated that a significant amount of x-rays are produced in the process. This was illustrated in an x-ray image produced via a collection of 20s images:

Finally, the authors performed a detailed study of the emission properties of sticky tape, and noted a striking correlation between the emission of x-rays and so-called ‘stick-slip’ events in the unspooling of the tape. The tape was unwound using a motor for which the force of unwinding could be measured, at a peel speed of 3 cm/s. It was noted that the emission is correlated very strongly with a sharp drop in the unwinding force; presumably this is a moment when a large section of tape suddenly comes loose from the spool.
X-rays with a peak photon energy of 15 keV were observed, and radio frequency emissions (much lower energy) were observed, also correlated with stick-slip.
This x-ray triboluminescence already looks to have possible application as an inexpensive, portable x-ray imaging system.
Though the explanation given above for the phenomenon seems reasonable, a detailed understanding of triboluminescence in this case (and in general) is still not available, in large part due to the complicated nature of the systems involved. Hopefully these results will shed some illumination on the phenomenon!
**********
* N.E. Harvey, “The luminescence of adhesive tape,” Science 89 (1939), 460-461.
** J.W. Obreimoff, “The splitting strength of mica,” Proc. R. Soc. A 127 (1930), 290-297.
*** V.V. Karasev, N.A. Krotova and B.W. Deryagin, “Study of electronic emission during the stripping of a layer of high polymer from glass in a vacuum,” Dokl. Akad. Nauk. SSR 88 (1953), 777-780.
Carlos G. Camara, Juan V. Escobar, Jonathan R. Hird, Seth J. Putterman (2008). Correlation between nanosecond X-ray flashes and stick–slip friction in peeling tape Nature, 455 (7216), 1089-1092 DOI: 10.1038/nature07378


I m having a hard time seeing this (wonderful!) effect used in place of the current x-ray stuff commercially, which in its cheapest form is a battery, a cathode ray tube, with a decent target.
Could you tune the frequency or something by changing the rate of pull away? I would see an apparatus as two wheels spinning against each other. You could certainly have a fun experimental set up with this though. Keeping the vacuum would be the thing. You would have mechanical things moving inside the container with their vibrations.
Markk wrote: “I m having a hard time seeing this (wonderful!) effect used in place of the current x-ray stuff commercially, which in its cheapest form is a battery, a cathode ray tube, with a decent target.”
It may have uses in developing countries where power sources are scarce or remote regions where carrying a bulky battery isn’t convenient. Your point is well-taken, though; as a theorist, I have a hard time estimating where a need might be filled.
“Could you tune the frequency or something by changing the rate of pull away?”
Possibly; the paper seems to have kept things going at a single rate of peeling, and doesn’t address whether any changes arise for different pull rates.
Interesting article. As of Markk’s question, changing the frequency would imply changing the energy level, since Plank’s constant and E=hv demands it. Changing the pull rate may require more mechanical energy and this may in turn imply more conversion into electrical energy. However, only the rate of change of electrical (static electricity) energy is more. Annihilation wise, more photons (rate) will be produced in a comparable time. For these photons to have more/less energy (and hence freq tuning), they need to arise from a greater energy quanta, which may seem likely if the rate of change of distance between the plates are rapid, forcing these particles to collide from a greater distance.
X-rays produced by Nanoparticles
It is unlikely the X-rays are produced by tearing electrons off glass or aluminum substrate. Electrons are more tightly bound to atoms than atoms are bound to each other. Instead, the tearing action likely forms nanoparticles (NPs) comprised of atoms of the adhesive. The X-rays therefore are somehow produced from otherwise neutral NPs.
Indeed, the X-rays in triboluminescence are similar to those formed in static electricity by the early Greeks when amber rods were rubbed by cloth. See http://www.nanoqed.net at links “A Unified Theory of Electrification in Natural Processes” and “Natural Electrification.”
In the production of X-rays by sticky tape, atoms in the adhesives before tearing are not under electromagnetic (EM) confinement and have full thermal kT energy. But in NPs, the atoms are under EM confinement at vacuum ultraviolet (VUV) levels that by quantum mechanics (QM) are restricted to vanishing kT energy. Therefore, as NPs form the atoms have excess kT energy beyond that allowed by QM. But the specific heat of NPs at high EM frequencies also vanishes, and therefore the excess in kT energy cannot be conserved by an increase in temperature.
Instead, conservation proceeds by the quantum electrodynamics (QED) induced up-conversion of kT energy to the EM confinement frequencies of the NPs at VUV levels. Each NP in the many formed therefore emits VUV electrons leaving behind positive charged molecules on both sticky and substrate sides, i.e., a film of adhesive always remains on the substrate. The VUV electrons are then accelerated to high velocities in the vacuum space by the positive charged adhesive on the tape and substrate film. Upon deceleration, the high energy electrons decelerate to produce the X-rays that enable radiographs of human fingers.
Triboluminescence is a variant of static electricity, but neither rely on tearing of electrons from atoms. Einstein showed by the photoelectric effect that EM and not mechanical energy is required to remove an electron from an atom. It is now an old wives tale that one can rub electrons off a material.
Thomas wrote: “It is unlikely the X-rays are produced by tearing electrons off glass or aluminum substrate. Electrons are more tightly bound to atoms than atoms are bound to each other.”
I’m not quite sure what your point is. Charge separation in the peeling process is readily observable with a pair of pieces of Scotch tape. Somewhere in the process, an electron is being pulled from a surface. And, it may not be what you’re saying, but the x-rays are not hypothesized to be created by the ‘tearing’ process, but rather by the recombination process.
“Einstein showed by the photoelectric effect that EM and not mechanical energy is required to remove an electron from an atom. It is now an old wives tale that one can rub electrons off a material.”
If you want to get very technical, all mechanical processes are really EM processes, as that is the force underlying all ordinary mechanical activity. And I’m not sure why you’re discussing ‘old wives tales’. It is an oversimplification to talk about ‘rubbing’ electrons off a material, but again it is readily shown that electrons get separated.
Skullsinthestars wrote: “it is readily shown that electrons get separated” and “all mechanical processes are really EM processes.”
The fact that electrons get separated does not mean they were separated by Mechanical processes – tearing, rubbing, or splitting materials, etc. Again, Mechanical processes cannot separate electrons from atoms.
My point is that the NPs that form in the peeling of tape or crushng of Lifesavers, or ….. produce the EM radiation that gives the impression that this occurred by Mechanical processes. The EM radiation is produced by QED induce EM radiation. See http://www.nanoqed.net
In the peeling of tape, the EM radiation from NPs produces positve charges on both the adhesive on both tape and substrate, the free electrons moving to the opposite positve charged surface and upon collision produce the X-rays.
As you say, “it is not possible to to prove that mica sheets, or…Scotch tape are oppsitely charged as a whole when pulled apart.” Opposing charges are likely local as shown in your second sketch of charge distributions.
The important point is the NPs produce a transient burst of VUV radiation that removes electrons from both the adhesive on the tape and the substrate. Electrons then move in the local electric field to the opposite positive charged surface that upon collision produce the X-rays by Bremsstralung. But the separation of charge occurs by EM and not Mechanical processes.
Similaly, the VIS light observed is produced by the VUV photons exciting fluorescent molecules in the adhesive. The fluorescent molecule in Wintogreen Lifesavers is methyl salicylate.
Perhaps, you can help me out to close this discussion.
I don’t know and cannot find out what the electron rich and fluorescent molecules are in Scotch tape necessary to produce the X-rays abd VIS light, repectively. Can you find this data?
That’s pretty sweet…
On the X-rays from Scotch Tape, please read my Press Release and paper.
http://www.prlog.org/10173358-nanoparticles-that-form-in-peeling-scotch-tape-produce-rays.html
Cool!
Well, if I ever have to do gift wrapping in a high vacuum, I guess I am going to need some protection. Of course, if I am gift wrapping in a vacuum, x-rays are about the least of my worries. Both fascinating and intriguing, and more then a little down right cool,
Thank you!
Cool! Though I do understand that x-ray in massive quantity is a health risk but knowing that x-rays can be produced and observed in simple ways is great.
Indeed!
Pingback: Atomic Tape X-Rays from Scotch Tape | Atomic Tape
Pingback: Barkla shows that x-rays have polarization (1905) | Skulls in the Stars
Pingback: Scientists crack the case of "screeching" Scotch tape - Copy Invisible News
Pingback: Scientists crack the case of “screeching” Scotch tape - Tech AI Verse
Pingback: اكتشف العلماء سر "صوت الصرخات" لشريط لاصق سكوتش
Pingback: Scientists crack the case of “screeching” Scotch tape – The TechBriefs
Pingback: Scientists Crack The Case Of "screeching" Scotc… – Patinko
Pingback: 科学家破获透明胶带“刺耳”的案例 - Mandarinian
Pingback: Scientists crack the case of “screeching” Scotch tape
Pingback: Scientists crack the case of “screeching” Scotch tape – City Wind News
Pingback: Scientists crack the case of “screeching” Scotch tape – ISE NEWS
Pingback: Научници откривају случај "шкрипања" селотејпа - Nezavisni sindikat prosvetnih radnika Vojvodine -