This result came out a few months ago, and I’ve been looking for the time to write about it ever since: in a paper published in the June 5 issue of Science, scientists reported the discovery of the first natural quasicrystal!
Of course, in order to get excited about this result, one needs to know what a quasicrystal is! In this post, we’ll take a look at what we mean by the terms ‘crystal’ and ‘quasicrystal’, and then explain why the discovery of a natural quasicrystal is significant.
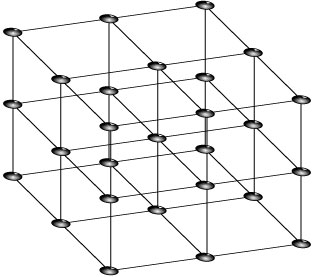
A crystal is a solid whose constituent atoms or molecules are arranged in a regular, repeating structure. For instance, a simple cubic crystal has atoms arranged on a cubic grid; a part of a cubic crystal is illustrated below:
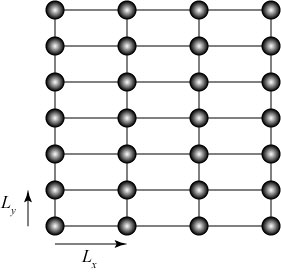
Such a structure is said to be periodic; moving in a straight line through the crystal, we regularly pass through points which are indistinguishable from our starting position. In a rectangular crystal, we can characterize this by introducing periods along the axes of the crystal; for instance, in a two-dimensional rectangular crystal can be characterized by the numbers and
,
Closely related to the idea of periodicity is the idea of long-range order. A system with long-range order is one for which distant locations have a strong relationship to one another. A periodic crystal clearly has long-range order: once the location of one atom of the crystal is specified, the position of every other atom is determined by the periodicity of the crystal.
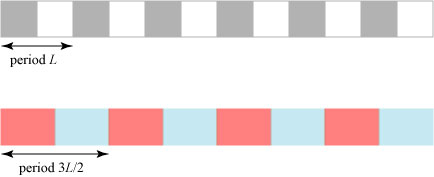
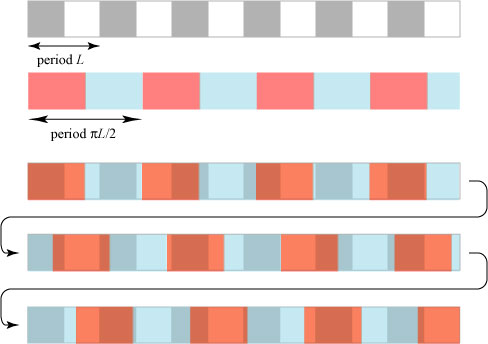
A quasicrystal is a structure that has long-range order but is nonperiodic. That is, there is a strong relationship between distant points of the structure even though the structure itself never repeats! To illustrate how this is possible, we consider a simple model of “one-dimensional crystals”, which we illustrate using colored blocks:
Here we have two different “crystals”: the white-gray crystal with period L and the red-blue crystal with period 3L/2; both are assumed to stretch off to infinity to the left and right. What happens if we create a new crystal by overlapping these two? We then get a crystal with period 3L:
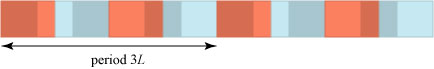
More generally, suppose we overlap two crystals, one with period L and one with period NL/M, where N and M are integers and the fraction N/M is reduced. One can reason that the combined pattern only repeats itself when N periods of the first crystal, or M periods of the second crystal, have passed. In this way we could make a pattern with a very large combined period, for instance if the second period is 999L/1000!
Let us now suppose that the period of the second crystal is , very close to 3L/2.
is an irrational number, which cannot be written in the form N/M. This means that a crystal made with the combined periods L and
will never exactly repeat itself1; a crude simulation of this is shown below:
This is what we might call a one-dimensional quasicrystal. It is nonperiodic — it never repeats — but it definitely has long-range order: knowing the two periods L and , and the point at which we begin, we can predict the behavior of the crystal at all points along its length.
This example is somewhat unsatisfying, because it depends upon having a ratio of periods exactly equal to an irrational number. Looking at higher-dimensional problems, such as a 2-D crystal or a fully 3-D crystal we may ask: can one construct a nonperiodic structure with long-range order in such cases? For quite some time it was generally assumed that long-range order and periodicity went hand-in-hand, but in 1974 mathematician and physicist Roger Penrose published a paper2 titled, “The Rôle of aesthetics in pure and applied mathematical research,” in which he showed that one can tile a surface nonperiodically using only a few shapes which are arranged according to a strict set of rules!
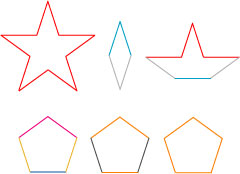
The shapes used in the original “Penrose tiling” are shown below:
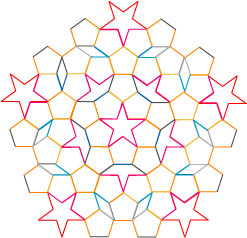
I have color-coded the pieces according to their allowable arrangements: light gray only next to dark gray, light yellow only next to orange, light blue only next to dark blue, and red only next to pink. An example of these pieces put into a pattern is shown below, hand-arranged by me in Adobe Illustrator:
I specifically made a pattern which would have 5-fold symmetry; that is, it looks the same if it is rotated through its center by 1/5 of a circle. One can in fact make even simpler nonperiodic tilings with only two pieces and an appropriate set of rules.
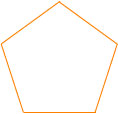
This tiling is nonperiodic, though this is difficult to prove rigorously. The pattern drawn above, however, gives a hint as to how this can be shown — the pattern drawn above is a symmetric pentagon, which means that it is simply a larger version of the original tile piece:
In order to create a periodic structure in two dimensions, we would need to be able to put our six pieces together in such a way as to make a shape that could be used to tile the entire plane without leaving any gaps. However, one can show that the six pieces are inevitably assembled into larger versions of themselves! These larger pieces can only be put together to make even larger pieces, and so on. By induction, we cannot combine them in a way to make a regularly repeating structure.
The Penrose tiling acts in a sense as an “anti-fractal”! Just as one can “zoom in” on a fractal structure and see that it is self-similar at a smaller scale, one can “zoom out” on a Penrose tiling and see that it is self-similar at a larger scale. This large scale similarity is also indicative of the long-range order of the tiling: the small pieces may be considered as sub-elements of a larger piece, and so on.
In three dimensions, similar nonperiodic tilings can be found, albeit ones which are much more complicated. The first physical quasicrystal was reported in 1984 by Shechtman and collaborators in a paper3 titled, “Metallic phase with long-range orientational order and no translational symmetry.” The term “quasicrystal” was coined soon afterwards in a theoretical paper describing the essential properties of such materials4.
Schechtman et al. studied alloys of Al-Mn which had been formed by quenching (rapid cooling). They discovered a quasicrystalline structure with icosahedral symmetry; that is, the crystal has a structure with symmetry properties comparable to a twenty-sided die:
(This particular die was auctioned at Christie’s last year and dates from 2nd century Rome.)
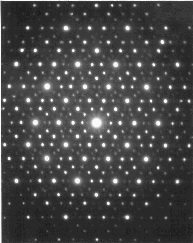
The symmetry properties of any crystalline or quasicrystalline structure can be determined by x-ray diffraction experiments. In the case of an icosahedrally-symmetric structure, one can observe diffraction patterns which are five-fold symmetric, three-fold symmetric, and two-fold symmetric; a five-fold symmetric pattern is shown below (via Cornell’s nice quasicrystals page):
The icosahedron axes of symmetry can be defined as follows:
- The icosahedron is five-fold symmetric when rotating about an axis which goes through a vertex and the center of the polygon,
- The icosahedron is three-fold symmetric when rotating about an axis which goes through a triangular face and the center of the polygon,
- The icosahedron is two-fold symmetric when rotating about an axis which goes through an edge and the center of the polygon.
No ordinary periodic crystal can have five-fold symmetry, though crystals occasionally “fake it” in a phenomenon known as “twinning“.
After the groundbreaking discovery of Shechtman and collaborators, a large number of quasicrystals were made artificially in the lab using a variety of different materials. All observed quasicrystals were synthetic, however, and no such materials had been found in nature; it was a logical next step for researchers to begin to hunt for natural versions of such crystals.
An organization known as the International Center for Diffraction Data has a large collection of diffraction data for a variety of materials, including some 9000 naturally occurring minerals. A search of the diffraction data for these materials, however, turned up no new quasicrystals, which led researchers to examine more exotic materials which have not been catalogued.
One candidate was the mineral khatyrkite, (Cu, Zn)Al2. This mineral is apparently very rare and is found in a region of the Koryak Mountains, northeast of the Kamchatka Peninsula in Russia. However, khatyrkite’s chemical composition is similar to that of known artificially-made quasicrystals, which made it a reasonable choice for study. In a paper published in Science in June of 2009, researchers L. Bindi, P.J. Steinhardt, N. Yao and P.J. Lu find that the sample of khatyrkite indeed possesses regions of icosahedral quasicrystalline behavior.
This discovery shows that quasicrystals can be formed naturally, and it significantly expands the list of naturally occurring mineral structures. The discovery of other natural quasicrystals could help researchers in determining other quasicrystalline structures and new techniques for constructing them.
One curious thought: given that khatyrkite is currently the only natural quasicrystal known, and samples of it are quite rare, is it now a valuable collector’s item? It would seem that, for the moment, it is one of the rarest materials on Earth…
******************************
1 It should be noted that this 1-D crystal will, however, come arbitrarily close to repeating itself an infinite number of times. For instance, because , the crystal will very nearly repeat itself after 100 periods of the second crystal, or after 471 periods of the first.
2 R. Penrose, “The Rôle of aesthetics in pure and applied mathematical research,” Bull. Inst. Math. Appl. 10 (1974), 266-271.
3 D. Shechtman, I. Blech, D. Gratias and J.W. Cahn, “Metallic phase with long-range orientational order and no translational symmetry,” Phys. Rev. Lett. 53 (1984), 1951-1953.
4 D. Levine and P.J. Steinhardt, “Quasicrystals: a new class of ordered structures,” Phys. Rev. Lett. 53 (1984), 2477-2480.
Bindi, L., Steinhardt, P., Yao, N., & Lu, P. (2009). Natural Quasicrystals Science, 324 (5932), 1306-1309 DOI: 10.1126/science.1170827



Pingback: Quasilinks for Quasicrystals, and Other Items « Galileo's Pendulum
do you know any artists who use crystal shapes in their artwork?
it’s pretty.
i don’t understand why the 2D example satisfies the necessary properties. we have this “anti-fractal” property, but i don’t see how this forces it to be aperiodic or to have long-range order. are you saying that as we zoom out and look for their inevitable formation of bigger copies of the same pieces, the pieces must be in a different formation each time? if not what is it that guarantees not periodic? if so, how do we know that happens? also, are you saying that there is only one way to glue these guys together? it doesn’t seem like that’s true. and without that, we certainly can’t say that given one piece we know what the entire rest of it looks like.
I didn’t really give a rigorous proof, which would be much more involved than I can do in a simple blog post! Perhaps the missing element is this: suppose you try arranging 100 pieces, of any kind, together to make an element that can then be assembled into a periodic pattern. This is, in essence, what periodicity would mean: some combination of piece would make a rectangle, equilateral triangle, or some other piece that could then be tiled infinitely. But, when we take those 100 pieces, we find that the best we can do is construct larger versions of the fundamental pieces, or combinations of them.
The problem has, in essence, been “reset”. We haven’t built a triangular or rectangular piece, but have only started the same problem over again on a larger scale. It follows, by induction, that any attempt to make triangles or rectangles (or hexagons) will fail on the larger scale, too! Once we’ve convinced ourselves that we can’t make a rectangle or equilateral triangle on a small scale, we know that we can’t do it on a larger scale. Since these are the only shapes that can be tiled periodically in a plane, we’ve essentially demonstrated that things don’t form a repeating pattern.
Pingback: These Mysterious Ultra-Rare Crystals Probably Formed In Outer Space | Gizmodo Australia