This post was written somewhat in conjunction with Jennifer Ouellette, who is posting at the same time at Cocktail Party Physics about Michael Faraday’s other classic Christmas lecture, The Chemical History of a Candle. Check it out, too!
Ah, Christmas! This is the time of year when I get together with family, exchange presents — and ruminate on the life and work of Michael Faraday.
It might seem an odd non sequitur to think about physicist and chemist Michael Faraday (1791-1867) during the holidays, but it is in fact highly appropriate. In addition to being a master scientist who helped unify the forces of electricity and magnetism, as well as magnetism and light, Faraday was a master lecturer. In 1825, in his new role as director of the laboratory of the Royal Institution, he instituted a yearly series of Christmas lectures to inform and educate the general public (especially young people) about science — these lectures are still ongoing today. Between 1827 and 1860, Faraday personally gave the Christmas lectures nineteen times, speaking on a variety of subjects.
Faraday giving a Christmas lecture in 1856 (source).
Unfortunately, records of only two of these lectures have survived: A Chemical History of a Candle (first given in 1848) and The Various Forces of Matter and their Relations to Each Other (given in 1859). Both of them are brilliant examples of clear scientific exposition, and in spite of how little was truly understood about physics and chemistry in the era they are still educational and even timeless. On this Christmas, I thought it would be worth looking at some aspects of Faraday’s fascinating views on the “forces of matter” in 1859.
Faraday was somewhat ideally suited to present popular lectures to the public. He came from a poor family and never received any formal schooling; at age fourteen he seemed destined for a relatively undistinguished life when he began a seven year apprenticeship with a bookbinder. The business gave him the opportunity to read from a variety of sources, however, and he was particularly inspired by the book Conversations in Chemistry by Jane Marcet. He ended up performing his own basic experiments based on the things he learned.
When nearing the end of his apprenticeship in 1812, Faraday got the opportunity to attend lectures by the esteemed chemist Humphry Davy. He took copious notes and performed follow-up experiments based on what he had learned. He later sent his volume of notes to Davy himself, impressing the man so much that Faraday was almost immediately hired to the Royal Institution as a Chemical Assistant. And the rest, as they say, was history — after many more trials and tribulations on Faraday’s part!
Faraday took the principles of lecturing seriously — his introduction to the Royal Institution allowed him to attend many talks, and he recorded detailed observations on what worked and what didn’t work when presenting to an audience. From a letter to his good friend Abbott, dated June 1, 1813, he writes:
The subject upon which I shall dwell more particularly at present has been in my head for some considerable time, and it now bursts forth in all its confusion. The opportunities that I have latterly had of attending and obtaining instruction from various lecturers in their performance of the duty attached to that office, has enabled me to observe the various habits, peculiarities, excellences, and defects of each of them as they were evident to me during the delivery. I did not wholly let this part of the things occurrent escape my notice, but when I found myself pleased, endeavoured to ascertain the particular circumstance that had affected me; also, whilst attending Mr. Brande and Mr. Powell in their lectures, I observed how the audience were affected, and by when their pleasure and their censure were drawn forth.
Faraday expounds on every conceivable detail important in giving a good lecture, among them: the size and shape of the lecture room, choosing the hour of the lecture appropriate for one’s audience, and the delivery of the lecture.
In order, therefore, to gain the attention of an audience (and what can be more disagreeable to a lecturer than the want of it?), it is necessary to pay some attention to the manner of expression. The utterance should not be rapid and hurried, and consequently unintelligible, but slow and deliberate, conveying ideas with ease from the lecturer, and infusing them with clearness and readiness into the minds of the audience. A lecturer should endeavour by all means to obtain a facility of utterance, and the power of clothing his thoughts and ideas in language smooth and harmonious and at the same time simple and easy.
Faraday puts these observations to good use in his Christmas lectures; even from reading them it is clear that they must have been delightful to behold. His lecture The Various Forces of Matter and their Relations to Each Other centers upon a theme that was central to Faraday’s entire career: the connections between the fundamental forces of nature.
In modern physics, it is essentially taken for granted that all of the forces of nature — gravity, electromagnetism, the strong nuclear force and the weak nuclear force — are all different manifestations of a single fundamental force. In Faraday’s time, the two nuclear forces were not even known, but the 1800s were nevertheless an era of unification — and Faraday was in the center of it. In 1831, Faraday rocked the scientific world by demonstrating that one could derive electricity from magnetism, completing a “circuit” that had been opened 11 years earlier with the discovery that magnetism could be generated by electricity. This would have been an accomplishment to secure his name in the history books, but he would achieve much more success: in 1845 Faraday demonstrated a connection between light and magnetism, paving the way for a complete electromagnetic theory of light. Faraday was perhaps the first true “unified field theorist”, even attempting in 1851 to experimentally demonstrate a connection between electricity and gravity!
So for his Christmas lecture of 1859, it was quite natural for Faraday to lay out what was known in his time of the fundamental forces of nature, and their known relationships. The lectures, delivered over a course of six days, were:
- The force of gravitation
- Gravitation — cohesion
- Cohesion — chemical affinity
- Chemical affinity — heat
- Magnetism — electricity
- The correlation of the physical forces
The list of forces, as enumerated above, are quite different from the fundamentals of today. Faraday highlights gravitation, cohesion (the attachment of atoms to similar atoms), chemical affinity (the interaction of atoms of different types with one another), heat, magnetism and electricity as the fundamental forces of note. It is interesting to see “heat” treated as a fundamental force, as we today know it to be the macroscopic manifestation of the random microscopic motion of atoms. Up until the mid 19th century, however, heat was envisioned as a fluid, known as caloric, and the properties of this fluid were considered distinct from those of, for instance, electricity.
The introduction to Faraday’s first lecture is a great example of his very personable, almost conversational, style of lecturing:
It grieves me much to think that I may have been a cause of disturbance in your Christmas arrangements, for nothing is more satisfactory to my mind than to perform what I undertake; but such things are not always left in our own power, and we must submit to circumstances as they are appointed. I will to-day do my best, and will ask you to bear with me if I am unable to give more than a few words; and as a substitute I will endeavour to make the illustrations of the sense I try to express, as full as possible; and if we find by the end of this lecture, that we may be justified in continuing them, thinking that next week our power shall be greater, —why then, with submission to you, we will take such course as you may think fit,—either to go on or discontinue them : and although I now feel much weakened by the pressure of illness (a mere cold) upon me, both in facility of expression and clearness of thought, I shall here claim, as I always have done on these occasions, the right of addressing myself to the younger members of the audience,—and for this purpose, therefore, unfitted as it may seem for an elderly infirm man to do so, I will return to second childhood and become, as it were, young again amongst the young.
Faraday begins his illustrations in a wonderful manner, illustrating force by considering different ways in which a piece of paper, propped upright, may be knocked over. A string dragged across the top of the paper will exert a force to pull it over, but a very different force can be applied by electrifying a piece of shell-lac and using its electrical attraction (though not yet described as such) to tip it. This simple illustration masks a bigger idea that Faraday gently leads his audience to — similar effects can be achieved by seemingly very different physical methods! It becomes almost natural, after Faraday completes his series of experiments, to believe that such forces much be connected in a very deep way.
We are not to suppose that there are so very many different powers; on the contrary, it is wonderful to think how few are the powers by which all the phenomena of nature are governed.
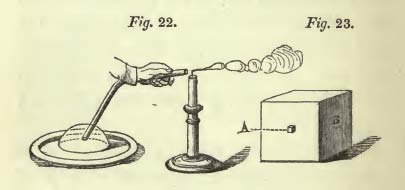
The lectures are all interconnected in a very clever way. Gravity is discussed in detail first, and it is used as a springboard, of sorts, to compare and contrast the behavior of the other forces of nature. Gravity’s fundamental nature is the simple experimental fact that all objects with mass “gravitate”, whether in solid, liquid, or gas form. Faraday illustrates this beautifully by generating a gas of (heavier than air) carbonic acid; this invisible gas, which can be held in a beaker, can be poured into another beaker on a balance and tip the scales, as illustrated in the printed version of Faraday’s lecture:
This simple demonstration is typical of those that Faraday presents during the course of his lectures. He conveys a very simple idea — that all matter, regardless of its state, experiences gravity — in a very visual and dramatic way.
From gravitation, Faraday moves on to the force of cohesion — the force by which matter of a certain type is bound to itself, for instance in a solid chunk of lead. Faraday demonstrates the almost magical properties of cohesion by demonstrating how a piece of lead may be broken into two pieces, and then reassembled with no other power than lead’s natural tendency to bind to itself:
I have here some pieces of lead which I melted this morning for the sake of making them clean. Now these pieces of lead hang together by the attraction of their particles, and if I press these two separate pieces close together, so as to bring their particles within the sphere of attraction, you will see how soon they become one. I have merely to give them a good squeeze, and draw the upper piece slightly round at the same time, and here they are as one, and all the bending and twisting I can give them will not separate them again ; I have joined the lead together, not with solder, but simply by means of the attraction of the particles.
Highlighted by the above passage, another aspect of Faraday’s lectures that are so effective is his ability to see remarkable phenomena in the most seemingly mundane objects. He truly does return to a “second childhood”, and this childlike enthusiasm brings wonder to everything he touches.
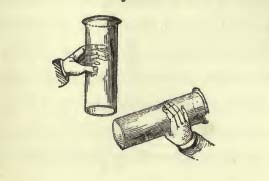
From a detailed discussion of cohesion, Faraday moves on to “chemical affinity”. He very cleverly refers back to experiments of the previous day to introduce the new ideas — a strategy I inadvertently discovered for my own lecture style! He refers back to the properties of cohesion by demonstrating how a soap bubble strongly compresses the air within it, so much so that the air escaping from the bubble can blow out an open flame (leftmost figure below):
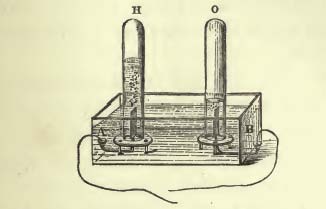
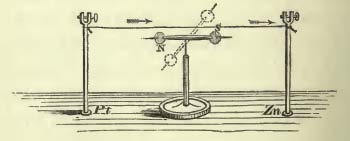
When we get to a discussion of the force of “chemical affinity”, the tendency of atoms of different type to join together, Faraday really enters his element! Much of his efforts focus on the decomposition of water (H2O) into its constituent elements, two atoms of hydrogen and one atom of oxygen. This relationship — two hydrogen for every single oxygen — is illustrated beautifully when he breaks up water using electricity! With the electrical poles submerged in water, he puts closed chambers above them to trap the escaping gasses. He finds that twice as much water has been pushed out of one tube as compared with the other, suggesting twice as much gas has been released into it:
(This picture seems to have a typo in it, in that it shows “O” having more water expelled than “H”, where they should be reversed.)
This use of electricity to decompose chemical affinity is not accidental: it again illustrates, and is referred to later, that one force can be used to subvert another, showing a connection. In this case, electricity can be used to break down chemical affinity, showing a connection.
From chemical affinity Faraday next lectures on “heat”, of which we hardly need to say much. Heat can be used to speed chemical reactions, including the combustion of materials, and electricity can of course produce heat itself. In this lecture, Faraday gives another charming demonstration, however, worth noting for its visual impact. As hydrogen gas is lighter than air, it floats upwards, and can be caught in an inverted glass. In a manner, then, it acts much like an “upside down liquid”, and can be poured from one upside down glass to another:
The presence of the gas can be confirmed with a simple match, as hydrogen is combustible. With the aid of a match, it will combine with the oxygen in the air to produce, of course, water.
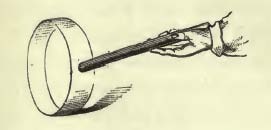
From heat, Faraday comes at last to his star subject: “electricity — magnetism”, and the relations thereof. He spends time demonstrating the existence of positive and negative electric charges via friction rods (shell-lac rubbed with flannel, or glass rubbed with silk), and even performs an experiment that has now morphed into a scientific toy! With a charged rod, a round strip of paper can be caused to roll along a surface, or even lifted up into the air:
This is the same principle that is used in the “FunFly Stick”, sold at the very clever “Grand Illusions” site:
The demonstration of electricity serves to highlight the large differences in the behavior of magnetism. Not only is magnetism in general a much stronger force, but it is much more persistent (not easily discharged as electricity is), and seems to perform its action at opposite “poles” of a magnet, which attract/repel in opposite sense.
But there is a connection, as laid out in Faraday’s concluding lecture, “The correlation of the physical forces”. Here he talks about all the ways in which one of the seemingly fundamental forces described can be converted into nearly any other one. For instance, one can use chemical means to produce electricity, or electricity to break down chemical affinity; also, as we have noted, one can demonstrate clearly the connection between electricity and magnetism. He reproduces Oersted’s classic experiment, in which a magnetic compass needle can be deflected by an electric current:
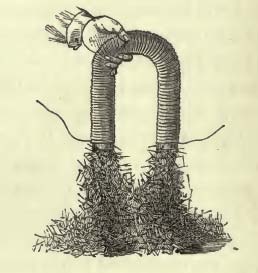
Much more dramatically, he shows that, by coiling a wire repeatedly around a U-shaped piece of iron and running a strong current through it, one can make a powerful electromagnet:
The really wonderful thing about Faraday’s lecture is that, by the end of the series, he has guided us gradually through simple demonstrations of a variety of physical phenomena and allowed us to, almost on our own, come to the conclusion that the forces of nature are intimately connected. As Faraday notes,
What then can surpass these evidences of the change of chemical force into electricity, and electricity into magnetism? I might show you many other experiments whereby I could obtain electricity and chemical action, heat and light from a magnet, but what more need I show you to prove the universal correlation of the physical forces of matter, and their mutual conversion one into another?
What indeed? Faraday acts more as a guide than a lecturer, and at the end of the lectures the journey seems eminently worthwhile. He concludes,
I hope that the insight which you have here gained into some of the laws by which the universe is governed, may be the occasion of some amongst you turning your attention to these subjects; for what study is there more fitted to the mind of man than that of the physical sciences? And what is there more capable of giving him an insight into the actions of those laws, a knowledge of which gives interest to the most trifling phenomenon of nature, and makes the observing student find
“tongues in trees, books in the running brooks,
Sermons in stones, and good in everything”?
I often wonder if Faraday’s lectures actually inspired any young people to go into science, and whether any of those young people went on to make important contributions. In any case, Faraday’s lectures can still serve as an inspiration for future scientists, and even current scientists such as myself. There is a lot to learn from Faraday’s lecture style, his ability to break down complicated subjects into simple demonstrations, and his genuine enthusiasm and love for the subjects.
Merry Christmas, and Happy Holidays!
*******************
You can read Faraday’s lectures for yourself on archive.org. You can also buy a copy at Dover Publications.





Pingback: Two good blogs | OMY
Pingback: Christmas with Faraday: The Chemical Record of a Candle – Pf Friend
Pingback: Faraday introduces the Forces of Nature | When I Discovered Your Words