![]()
I thought I’d step out of my comfort zone and specific field of expertise for once and do a post on some interesting quantum optics. In a June issue of Physical Review Letters, an Israeli research group experimentally demonstrated the ability to store and retrieve optical images in an atomic vapor using so-called ‘electromagnetically induced transparency’, a purely quantum-mechanical effect. Researchers have previously demonstrated the ability to ‘freeze’ light pulses in an atomic medium, but this is the first time to my knowledge that a structured two-dimensional image has been given the same treatment. To fully describe the research and its significance, however, we need to say a little bit about how atoms and molecules absorb and emit light.
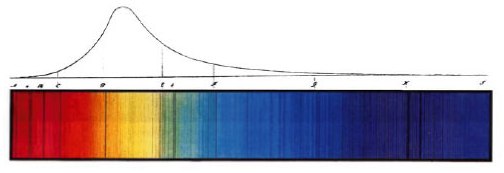
The best way to do this is to look at a specific, simple example, the hydrogen atom, and stick to the simplest model of said atom, due to Bohr. We’ve talked about Bohr’s 1913 model of the hydrogen atom before, and a short summary is worth repeating. Physicists of the early 1900s had determined that atoms only emit/absorb radiation at certain discrete frequencies, and this phenomena could not be explained by the classical physical theory of the time. An example of this special absorption was discovered by Fraunhofer in 1814 when he measured the spectrum of light from the Sun:
One can see from Fraunhofer’s hand-drawn sketch that the Sun emits light at nearly all frequencies, except for isolated ‘special’ frequencies where the spectrum appears dark. These special frequencies are the frequencies at which the atoms of the sun absorb light, though an explanation for their origin was not found for nearly a century.
Bohr solved the problem by proposing a model for the atom which departed completely from the known physics of the time. He proposed several postulates for the behavior of the electrons in the atom:
- Electrons can only travel in stable orbits which possess quantized angular momentum, i.e. the orbital angular momentum of the electron satisfies the formula:
,where h is Planck’s constant, introduced in the study of blackbody radiation, and n = 1,2,3, and so forth. This in turn suggests that the electron travels only travel at certain orbital distances from the nucleus.
- An electron can only give up its energy “all at once” by jumping from a higher n-state to a lower n-state. The frequency
of the photon (light particle) released in jumping from level 2 to level 1, for instance, is given by
, where
is the energy of the particle in the n = 1 state.
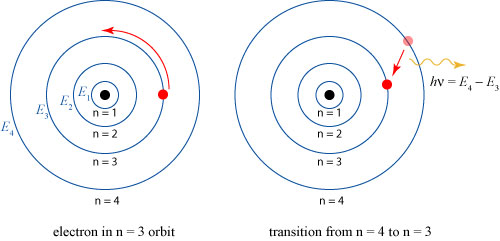
This is a lot of words, but the simple significance of the model is illustrated below:
The electron can only reside in certain ‘allowed’ orbits, labeled by the index . An electron transitions between the different orbits by releasing/absorbing energy in the form of a photon (light particle). The frequency (color)
of the photon is simply the difference in the energy levels.
Bohr’s model answered many scientific questions in its time and put science on the road to quantum mechanics, though the model itself is oversimplified and, in the strictest sense, incorrect. Electrons do not orbit the nucleus like a classical particle; rather, they possess wave properties of their own and the electron ‘orbit’ is really a stretched-out electron wave that surrounds the nucleus. These wave properties are crucial for understanding electromagnetically induced transparency, and we will return to them in a moment.
For now, we note that all individual atoms and molecules behave similarly to the hydrogen atom: they possess numerous (in fact, infinite) discrete energy states in which they can reside, and they absorb/release photons by transitioning between these states. Typically these states are illustrated using an energy-level diagram, as illustrated below for hydrogen:
The vertical axis represents the energy of the particular quantum state. The horizontal axis is used to label other differences between quantum states. In the case of hydrogen, a unique state of the electron is not just distinguished by the energy of the electron, but also by its angular momentum . (This is something else Bohr got wrong, as he essentially identified the quantum number
with the angular momentum.) There are multiple angular momentum states (
) with the same energy (
); for instance, for
there are four angular momentum states. The dense collection of lines near the top of the diagram represents an infinite number of energy states which lie between the
level and a liberated electron
.
Here’s where things get weird. Because an electron has wavelike properties, it can actually reside in multiple energy levels simultaneously. This state of affairs can last until an ‘observer’ makes an explicit measurement of the energy of the electron, at which point it randomly ‘chooses’ one of the levels to stay in. This behavior is illustrated most famously (or infamously) in the so-called Schrödinger’s cat paradox. In this thought experiment, a cat is placed in a box containing a poison capsule whose opening is dictated by the behavior of a single atom. The cat is supposedly simultaneously alive and dead until an observer opens the box to determine the animal’s health.
If this doesn’t seem to make much sense to you, you’re in agreement with most physicists. Schrödinger himself came up with the paradox to illustrate the seemingly nonsensical nature of quantum mechanics. However bizarre the consequences, quantum mechanics as a theory works, and most scientists accept the basic principles as correct. (It is technically unfeasible to perform Schrödinger’s experiment with an actual cat, as the ASPCA will be happy to learn.)
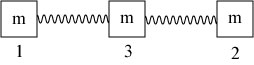
We now come back to the idea of electromagnetically induced transparency, which is the physical mechanism used to slow down and/or freeze light beams. To give an analogy, let’s play with some springs! Suppose we have a series of 3 equal masses connected by springs as illustrated below:
The springs are assumed to be identical as well; the numbering of the springs is in anticipation of the EIT discussion to follow. Suppose we excite one of the outer masses, say 1, with an oscillatory motion. The spring attached will apply a force to mass 3, which in turn will apply a force to mass 2. In the end, all three masses end up moving.
What happens if we excite the outer two masses simultaneously, with equal strength? There are two distinct ways we can do this: the two masses can move in the same direction, or in opposite directions, as animated below:
Symmetric motion (1 + 2):
Asymmetric motion (1 – 2):
All internal oscillations of this three spring system can be reduced to some combination of the two excitations shown above; we label the two types of motion “(1+2)” and “(1-2)”. In the (1+2) configuration, the motion of the outer two masses couples via the springs into motion of the third mass. In the (1-2) configuration, however, the effect of the outer two masses completely cancel, leaving the third mass unmoving. This behavior is the equivalent to what is known in quantum optics as a dark state, and the phenomenon of electromagnetically induced transparency is related to the existence of dark states in quantum systems.
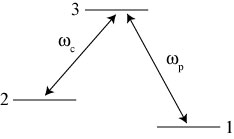
We would like to draw an analogy between our spring system and an atomic system with three energy levels arranged as illustrated below1:
This system is usually referred to as a ‘lambda’ system, because of its similarity to the Greek letter . The energy difference between levels 1 and 3 is
, while the energy difference between levels 2 and 3 is
. We assume that a physical constraint on the system prevents transitions directly from level 1 to level 2.
Suppose we illuminate this ‘lambda’ atom with two different lasers, a ‘probe field’ oscillating at frequency and a ‘coupling field’ oscillating at frequency
. The atom likely starts in one of the two lower energy states, 1 or 2, but eventually absorbs a photon of appropriate energy and ends up in the higher energy state 3.
Here’s where it gets interesting. The atom inevitably releases its energy in the form of another photon and decays back down to a lower energy state. Because of the wave nature of the atom, however, it will decay down into a superposition state, just like our earlier spring system. Roughly speaking, when the atom decays, it either ends up in the (1+2) state or the (1-2) state.
In analogy with our spring system, an atom in the (1+2) state can still excite state 3, in which case the atom absorbs a photon and goes back into the 3 state. But if it ends up in the (1-2) state, it has no ability to get back into state 3. The effect of the coupling laser trying to push the atom from state 2 to state 3 is canceled completely by the effect of the probe laser trying to push the atom from state 1 to state 3. Kind of like the losing team in a game of dodgeball, eventually every atom in our system will end up in the dark state (1-2) and be ‘eliminated’ from any possibility of absorbing light. All the atoms get stuck in the (1-2) state; this process is known as coherent population trapping, and was first observed in 1976.2
But if the atoms cannot absorb light, they are effectively invisible to the light source: the medium becomes transparent to the probe field. This is the phenomenon of electromagnetically induced transparency: the interaction of the two light fields in the medium has made the medium transparent.3
With EIT (roughly) explained, we can now move much faster towards explaining the ‘freezing’ of images! It is hopefully somewhat clear from the discussion above that the EIT effect depends strongly on the frequencies of the coupling and probe laser matching the energy transitions of the atom. This in turn means that the refractive index of the medium depends strongly on the frequencies of the lasers. However, as we’ve discussed previously in an optics basics post, the group velocity of a wave depends upon the variation of frequency with wavelength or, equivalently, the variation of refractive index with wavelength:
.
The (calculus-based) quantity , which is a measure of how fast the index
changes as we change
, is huge for a medium undergoing EIT, which means that the group velocity is tiny: probe waves undergoing EIT move at a very slow velocity in the medium.
This was demonstrated dramatically in 1999 when a research group slowed light down to 17 m/s in an ultracold gas of sodium atoms!4 As was noted in the press at the time, this is a reduction of the speed of light to about that of a fast-moving bicycle!
Only two years later, very nearly the same research group5 demonstrated that they could ‘halt’ a light pulse traveling in an EIT medium by switching off the coupling laser sharply. With the coupling laser off, the field becomes trapped in a complicated way within the atoms: the atoms do not simply absorb the light, but ‘record’ the wave properties of the light field within their own atomic wavefunctions. When the coupling field is turned on, the field is released from the atoms and travels onward as if nothing has happened.
There is a lot of interest in making optical computers, i.e. computers which use light instead of electrons to manipulate, transmit and store data. The ability to slow down and store light fields, as the previously mentioned experiments showed, is a big step towards making such computers reality. The previous experiments only slowed/trapped/stored a one-dimensional pulse of light; the next interesting question to ask is whether images (pictures) can also be stored.
A group from the University of Rochester6 managed to slow two-dimensional images in a highly dispersive Cesium vapor in 2007. The first experiment to actually freeze and store a two-dimensional image, however, was reported just this June in Physical Review Letters by an Israeli group. The researchers transmitted simple images through a warm (52° C) gas of 87Rb and managed to store them for up to 30 microseconds (30 millionths of a second). This may not seem to be very long, but it is a delay comparable to the amount of time it would take the light to freely travel through the gas. It is further remarkable because the gas is warm: the atoms are moving around quite rapidly, but recognizable images could still be recovered.
The motion of the gas presented unique problems for the researchers. The gas spreads out over time, taking the stored light signal with it. The diffusion of the gas results in a diffusion of the images, so that over a long enough storage time they become unrecognizable. I was unable to get the original images for the paper, but what follows is a crude simulation of this effect:
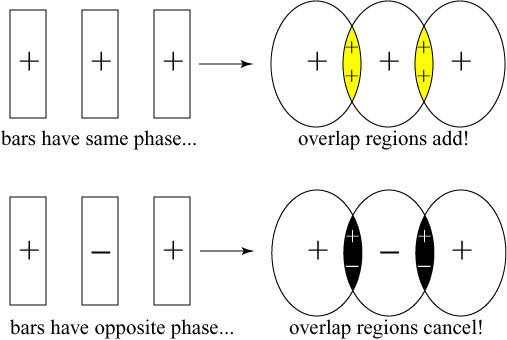
The result is significantly worse in the actual data. To compensate for the blurring of the images, the researchers used a technique which is standard in optical lithography, known as phase-shifting. In the researchers’ original experiment, the three image bars had exactly the same phase, but in a revised experiment they shifted the phase of adjacent bars by 180°. How does this help? When the images begin to blur, and parts of the adjacent bars overlap, their contributions cancel each other out. This is schematically illustrated below:
For simple images such as the three bars, this method keeps the images well-separated, but it is not clear how to apply it in the case of extremely complicated pictures. As a side-effect, though, the improvement in the phase-shifted images versus the non-shifted images demonstrates that the phase of the optical images is indeed being stored in the medium.
Let me conclude on a little personal anecdote. Several years ago, my thesis advisor was explaining to me how the military was very interested in producing ‘laser-proof’ goggles. The idea would be to have goggles which become opaque before the laser-light reaches the eyes of an aircraft pilot; this could protect against an enemy intentionally using lasers as a blinding weapon. (Which is a technology the U.S. military has developed themselves.) My advisor and I had a good chuckle about the idea: in order to make goggles of this sort, one would need to have electronics that, in essence, respond faster than the speed of light. At that time, no material was known that could slow light to such an extent to make this idea feasible.
Shows what we knew! These experiments in slowing and stopping light have made impossible technologies become possible and, perhaps, eventually reality.
*******************************************
Shuker, M., Firstenberg, O., Pugatch, R., Ron, A., Davidson, N. (2008). Storing Images in Warm Atomic Vapor. Physical Review Letters, 100(22) DOI: 10.1103/PhysRevLett.100.223601
Notes:
1 Here I get a little explanatory guidance from the excellent book by P.W. Milonni, Fast Light, Slow Light and Left-Handed Light (IOP Press, 2005).
2 G. Alzetta, A. Gozzini, L. Moi and G. Orriols, “An experimental method for the observation of R.F. transitions and laser beat resonances in oriented Na vapour,” Nuovo Cimento B 36 (1976), 5-20.
3 There are many more details to be attended to and more conditions which must be satisfied in order for EIT to succeed; the review article, S.E. Harris, “Electromagnetically induced transparency,” Phys. Today, July (1997), 36-42, gives explanations and early references.
4 L.V. Hau, S.E. Harris, Z. Dutton and C.H. Behroozi, “Light speed reduction to 17 metres per second in an ultracold atomic gas,” Nature 397 (1999), 594-598.
5 C. Liu, Z. Dutton, C.H. Behroozi and L.V. Hau, “Observation of coherent optical information storage in an atomic medium using halted light pulses,” Nature 409 (2001), 490-493.
6 R.M. Camacho, C.J. Broadbent, I. Ali-Khan and J.C. Howell, “All-optical delay of images using slow light,” Phys. Rev. Lett. 98 (2007), 043902.


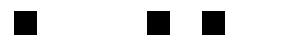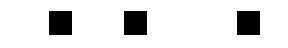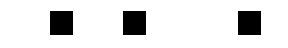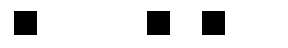
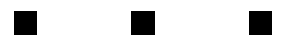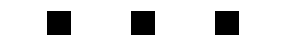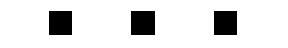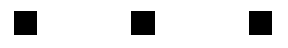


I read this paper when it was first published last month, but I don’t think I fully understood it or appreciated its implications, until after I read your post. Thanks for the detailed explanation.
At the end of the paper, the Israeli researchers suggest that “it should be possible to store more elaborate temporal shapes, thus storing three-dimensional information” and they allude to the possibility of storing “movies”. I suspect the diffusion of the atoms would make this rather unfeasible, although there may be some limited practical uses (not a full-length DVD!).
stuwat: Thanks for the comment! Yeah, I doubt one will be able to store significant amounts of information for significant periods of time due to diffusion. I’m sure this has some potential in fibre optics systems, though. I’m not so good at the application side of such technologies, but a 30 microsecond delay could be used to replace a lot of optical delay line.
Now I understand the implications and applications of the paper much better. Thanks for the clear description.
IronMonkey: I’m glad it made sense! As I said, this was significantly outside of my specialization, so the compliments are appreciated.
But you’re not having ftl here, are you.
You are just slowing it.
That is if ftl is defined as from a vacuum.
But it is a very interesting question.
As ftl is a ‘barrier’, just like the event horizon.
And by manipulating density and ‘internal motion’ we get ‘strange’ results.
In fact 🙂
Maybe I should just write ‘internal motion’?
As that seem to be ‘density’ 🙂