Michael Faraday (1791-1867), whom I’ve talked about numerous times, has a reputation as being a bit of a theoretical lightweight, namely because he had little formal mathematical training. In spite of this, however, he had an ability to think abstractly and, yes, theoretically about problems in a way that, when examined, is nothing short of amazing.
In my previous researches on Faraday, I came across a reference to an article he wrote in 1844 in volume 24 of Philosophical Magazine, pp. 136-144, “A speculation touching electric conduction and the nature of matter.” Faraday, already a distinguished and even famous scientist, shared some thoughts about the nature of atomic structure, based on the paucity of knowledge that was available at the time. His observations, though still off the mark according to current understanding, were remarkably forward thinking; furthermore, they provide a lovely snapshot of the ‘state of the art’ in 1844. Let’s take a look at his paper:
It is worth noting that extremely little was known about the structure of the atom in Faraday’s time; you can see my gallery of failed atomic models to see how confused the field was even fifty years later. Faraday summarizes the thinking of his contemporaries:
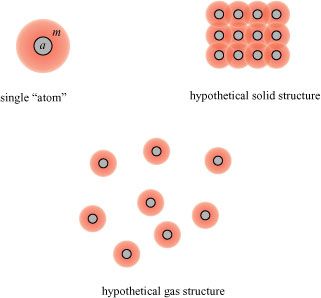
The view of the atomic constitution of matter which I think is most prevalent, is that which considers the atom as a something material having a certain volume, upon which those powers were impressed at the creation, which have given it, from that time to the present, the capability of constituting, when many atoms are congregated together into groups, the different substances whose effects and properties we observe. These, though grouped and held together by their powers, do not touch each other, but have intervening space, otherwise pressure or cold could not make a body contract into a smaller bulk, nor heat or tension make it larger; in liquids these atoms or particles are free to move about one another, and in vapours or gases they are also present, but removed very much further apart, though still related to each other by their powers.
In short, atoms were considered indestructible hard spheres, which we will label a, surrounded by a field of force labeled m; solids, liquids and gasses are distinguished by the separation of the spheres:
Some space must always be present between the spheres, even in solids, otherwise materials could not contract when cooled.
Faraday then shows incredible insight in pointing out that this picture of atoms is not an experimentally observed fact, but an assumption based on the facts which has not been directly proven:
The atomic doctrine is greatly used one way or another in this, our day, for the interpretation of phaenomena, especially those of crystallography and chemistry, and is not so carefully distinguished from the facts, but that it often appears to him who stands in the position of student, as a statement of the facts themselves, though it is at best but an assumption; of the truth of which we can assert nothing, whatever we may say or think of its probability. The word atom, which can never be used without involving much that is purely hypothetical, is often intended to be used to express a simple fact, but, good as the intention is, I have not yet found a mind that did habitually separate it from its accompanying temptations; and there can be no doubt that the words definite proportions, equivalents, primes, &c., which did and do express fully all the facts of what is usually called the atomic theory is chemistry, were dismissed because they were not expressive enough, and did not say all that was in the mind of him who used the word atom in their stead; they did not express the hypothesis as well as the fact.
In other words, the “atomic theory” explained nicely the existing experimental observations about chemistry, such as the law of definite proportions, but also brought with it a lot of unjustified baggage; namely, the picture of “atoms” as hard impenetrable spheres. Faraday then considers whether this “baggage” is itself justified, based on the facts available to him.
He begins by thinking about how the existing “atom” picture relates to the existence of electrical conductivity:
If the view of the consitution of matter already referred to be assumed to be correct, and I may be allowed to speak of the particles of matter and of the space between them (in water, or in the vapour of water for instance) as two different things, then space must be taken as the only continuous part, for the particles are considered as separated by space from each other. Space will permeate all masses of matter in every direction like a net, except that in place of meshes it will form cells, isolating each atom from its neighbours, and itself only being continuous.
Then take the case of a piece of shell-lac, a non-conductor, and it would appear at once from such a view of its atomic constitution that space is an insulator, for if it were a conductor the shell-lac could not insulate, whatever might be the relation as to conducting power of its material atoms; the space would be like a fine metallic web penetrating it in every direction, just as we may imagine of a heap of siliceous sand having all its pores filled with water; or as we may consider of a stick of black wax, which, though it contains an infinity of particles of conducting charcoal diffused through every part of it, cannot conduct, because a non-conducting body (a resin) intervenes and separates them one from another, like the supposed space in the lac.
Next take the case of a metal, platinum or potassium, constituted, according to the atomic theory, in the same manner. The metal is a conductor; but how can this be, except space be a conductor? for it is the only continuous part of the metal, and the atoms not only do not touch (by the theory), but as we shall see presently, must be assumed to be a considerable way apart. Space therefore must be a conductor, or else the metals could not conduct, but would be in the situation of the black sealing-wax referred to a little while ago.
But if space be a conductor, how then can shell-lac, sulphur, &c. insulate? for space permeates them in every direction. Or if space be an insulator, how can a metal or other similar body conduct?
It would seem, therefore, that in accepting the ordinary atomic theory, space may be proved to be a non-conductor in non-conducting bodies, and a conductor in conducting bodies, but the reasoning ends in this, a subversion of that theory altogether…
Faraday seems to be basing his reasoning on the idea that electricity is a fluid which is transmitted only through physical contact with a “material”, as it appears on the macroscopic level. Then the only thing that electricity can flow through is the assumed empty space between atoms, but this space appears as an insulator or a conductor, depending on the material.
Can you see the flaw in the argument? The reasoning seems sound, based on the assumptions made, but the major assumption itself is wrong. Electricity acts not as a fluid but is in fact carried by electrons, which can travel easily through empty space. The conducting/insulating properties of matter depend not on the properties of space but rather how freely electrons can migrate through the material in question. Of course, Faraday did not known about the electron, which would not be discovered until 1897, though speculation of its existence first arose even earlier than Faraday’s “speculations”.
Following up on this reasoning, Faraday further points out that there is no obvious correlation between conducting power and atomic density. One might assume naively that more atoms = more conduction, but this is not the case. Iron has a high atomic density, but low conductance compared to gold, which has a much lower atomic density. Faraday notes,
And all this happens amongst substances which we are bound to consider, at present, as elementary or simple. Whichever way we consider the particles of matter and the space between them, and examine the assumed constitution of matter by this table, the results are full of perplexity.
With the atomic model of Faraday’s time, one cannot make any predictions about conduction based on the “atomic structure”, which suggests that the system is incomplete or simply incorrect.
The most striking argument Faraday puts forth, however, concerns the densities of chemicals with similar constituents, and the paradoxes which seem to arise when interpreting them via the assumed atomic model. As a fun exercise, while reading Faraday’s argument, see if you can see what aspect of real atomic structure Faraday is suggesting:
Now let us take the case of potassium, a compact metallic substance with excellent conducting powers, its oxide or hydrate a non-conductor; it will supply us with some facts having very important bearings on the assumed atomic construction of matter.
When potassium is oxidized an atom of it combines with an atom of oxygen to form an atom of potassa, and an atom of potassa combines with an atom of water, consisting of two atoms of oxygen and hydrogen, to form an atom of hydrate of potassa, so that an atom of hydrate of potassa contains four elementary atoms. The specific gravity of potassium is 0.865, and its atomic weight is 40; the specific gravity of case hydrate of potassa, in such state of purity as I could obtain it, I found to be nearly 2, its atomic weight 57. From these, which may be taken as facts, the following strange conclusions flow. A piece of potassium contains less potassium than an equal piece of the potash formed by it and oxygen. We may cast into potassium oxygen atom for atom, and then again both oxygen and hydrogen in a twofold number of atoms, and yet, with all these additions, the matter shall become less and less, until it is not two-thirds of its original volume. If a given bulk of potassium contains 45 atoms the same bulk of hydrate of potassa contains 70 atoms nearly of the metal potassium, and besides that, 210 atoms more of oxygen and hydrogen. In dealing with assumptions I must assume a little more for the sake of making any kind of statement; let me therefore assume that in the hydrate of potassa the atoms are all of one size and nearly touching each other, and that in a cubic inch of that substance there are 2800 elementary atoms of potassium, oxygen and hydrogen; take away 2100 atoms of oxygen and hydrogen, and the 700 atoms of potassium remaining will swell into more than a cubic inch and a half, and if we diminish the number until only those containable in a cubic inch remain, we shall have 430, or thereabout.
So a space which can contain 2800 atoms, and amongst them 700 of potassium itself, is found to be entirely filled by 430 atoms of potassium as they exist in the ordinary state of that metal. Surely then, under the suppositions of the atomic theory, the atoms of potassium must be very far apart in the metal, i.e. there must be much more of space than of matter in that body: yet it is an excellent conductor, and so space must be a conductor; but then what becomes of shell-lac, sulphur, and all the insulators? for space must also by the theory exist in them.
There’s a lot of words in that argument, but it can be boiled down very simply. If we take a sample of potassium, and ‘magically’ add lots of oxygen and water to it to make hydrate of potassa, the sample gets much smaller! Adding atoms reduces the volume of the sample! Certainly the chemical forces could draw the atoms closer together, but only if they were appreciably separated to begin with. If an atom consists of a ‘hard impenetrable sphere’, the region around a single atom in a material must contain much more space than matter!
This argument is very much reminiscent of the crude statement that an atom consists almost entirely of empty space! We now know that an atom consists of an extremely small nucleus of matter surrounded by a quantum-mechanical cloud of electrons.
One curious note: Faraday refers to “an atom of water, consisting of two atoms of oxygen and hydrogen.” Two atoms of oxygen in water? This isn’t a typo, because Faraday refers to it again later. Was water imagined to be back then?
Returning to the nature of the atom, how did Faraday perceive the seeming ‘flexibility’ in the size of atoms? It would be over fifty years before Rutherford postulated the nuclear model of the atom and began the field of nuclear physics, but Faraday had his own ‘nuclear’ view of the atom:
If we must assume at all, as indeed in a branch of knowledge like the present we can hardly help it, then the safest course appears to be to assume as little as possible, and in that respect the atoms of Boscovich appear to me to have a great advantage over the more usual notion. His atoms, if I understand alright, are mere centres of forces or powers, not particles of matter, in which the powers themselves reside. If, in the ordinary view of atoms, we call the particle of matter away from the powers a, and the system of powers or forces in and around it m, then in Boscovich’s theory a disappears, or is a mere mathematical point, whilst in the usual notion it is a little unchangeable, impenetrable piece of matter, and m is an atmosphere of force grouped around it.
In many of the hypothetical uses made of atoms, as in crystallography, chemistry, magnetism, &c., this difference in the assumption makes little or no alteration in the results, but in other cases, as of electric conduction, the nature of light, the manner in which combine to produce compounds, the effects of forces, as heat or electricity, upon matter, the difference will be very great.
…
To my mind, therefore, the a or nucleus vanishes, and the substance consists of the powers or m; and indeed what notion can we form of the nucleus independent of its powers? all our perception and knowledge of the atom, and even our fancy, is limited to the ideas of its powers: what though remains on which to hang the imagination of an a independent of the acknowledged forces?
We’ll return to a discussion of Boscovich in a later post. The idea, however, is simple: instead of treating atoms as hard spheres of matter a which are coupled together by forces m, why not dispense with the spheres, which were only assumed to exist in Faraday’s time.
This view is quite bold, and seemingly accounts for the perplexing observations Faraday describes. An atom as a ‘center of force’ is flexible and could account for the unusual density effects in chemical compounds. The view that matter consists entirely of forces, and not “matter”, means that there exists no “space” between atoms.
Though this view is a great oversimplification of the reality of atoms — consisting of a nucleus which in turn consists of subatomic protons and neutrons which in turn consist of quarks — it is a striking attempt to come up with a simplified theory of matter and a nice philosophical attempt to remove what might be called the “metaphysical speculation” about the nature of matter. The notion that atoms are malleable bodies whose boundaries are only defined by the balance of forces with neighboring atoms is much closer to the current quantum-mechanical view of atomic structure than the ‘hard sphere’ theory that was prevalent at the time.
It is a fascinating historical footnote that Faraday uses the word “nucleus” to describe his “centers of force,” though the word nucleus has a very different meaning in its modern usage.
The response to Faraday’s work seems to have been somewhat mixed, though always respectful. In 1845*, a Dr. Hare made remarks critical of Faraday’s speculation. A reading of Hare’s work to me showed that he was not as clear a communicator as Faraday, as I had a hard time following his arguments. However, the main thrust seems to be encapsulated in the following statements:
It follows that the existence of an immense calorific and electrical power is undeniable. The materiality of these powers, or of their cause, is all that has been questionable. But, according to the speculations of Faraday, all the powers of matter are material; not only the calorific and electrical powers are thus to be considered, but likewise the powers of cohesion, chemical affinity, inertia and gravitation, while of all these material powers only the latter can be ponderable!!!
Thus a disinclination on the part of this distinguished investigator to admit the existence of one or two imponderable principles, has led him into speculations involving the existence of a much greater number. But if the calorific and electrical powers of matter be material, and if such enormous quantities exist in potassium, as well as in zinc and all other metals, so much of the reasoning in question as is founded on the vacuity of the space between the metallic atoms, is really groundless.
Hare seems to be arguing that, in Faraday’s system, there is no difference between matter and the forces, or “powers” of nature. By Hare’s reasoning, pure potassium has so much more space per atom than hydrate of potassa because each atom has so much more calorific and electrical power, which must themselves take up space. But then Faraday’s concerns about the space between atoms are rendered moot, because that space may be considered to be filled with material “power”.
Another criticism came from a Dr. Mayo (via Faraday: His Life and Letters), who seems to have questioned whether Faraday’s speculation really solves any problems about atomic structure, or just ignores them:
Your atmosphere of force, grouped round a mathematical point, is not, as other hypothetical expressions have been in the course of your researches, an expression linking together admitted phenomena, but rather superseding the material phenomena which it pretends to explain. It resolves, in fact, as it would appear to me, all matter into a metaphysical abstraction. For it must all consist of the mathematical point, and the atmosphere of force grouped around it.
You ought perhaps to carry your disposition to limit our real knowledge of things to effects and laws a little further, and apply it also to your own hypothesis. A mathematical point with an atmosphere of force around it, is in respect to the atmosphere of force an expression of certain effects. But what is the mathematical point?
Mayo is apparently questioning whether Faraday’s “centers of force” really answer any questions, or just replace one set of assumptions with another. This is a valid criticism; as noted, Faraday’s speculations seem to have pointed out some of the “missing links” in atomic theory (namely, electrons and nuclei), but his solution is replacing of one speculative structure with another.
Was Faraday just wasting his time, then? In other words, is it “pointless” (pardon the pun) to come up with new hypotheses to explain nature, in the absence of experimental evidence? This is a question which is extremely relevant today in string theory, where theoreticians are speculating again on the absolute nature of matter. Dr. Mayo’s introduction to his letter gives a nice point of view on this subject:
Believing that no analytical inquiry has ever been set on foot, without some preconceived hypothesis, I imagine also that theory and hypothesis never need interfere with the prosecution of an inquiry… Your discoveries, indeed, sufficiently show the value of hypothesis. For no man uses its language more successfully than you do, as the associating agent in your analytical inquiries. In this respect your intellectual operations supply a striking proof of the value of a vivid imagination in a philosopher.
In short, hypothesis and speculation are natural parts of the scientific process, and even necessary ones. An idea which gets people to think about a physical problem in a new and different way can be a stepping stone to a new discovery, even if the idea turns out in the end to be inaccurate.
***********************************************
* Dr. Hare, “On a recent ‘speculation’ by Faraday,” American Journal of Science and Arts 48 (1845), 247-252.



Thanks. I enjoyed reading that.
(I first read your blog when swansont linked to
levitation-and-diamagnetism-or-leave-earnshaw-alone)
Colonal: Thanks for the feedback!
Regarding the composition-of-water question, I finally managed to remember where I had read the passage that I had been thinking of. Originally, it appears, Dalton thought that one molecule of water was composed of one hydrogen atom and one oxygen atom (HO rather than H2O). Quoting from the “1803” section of Asimov’s Chronology of Science and Discovery (1988):
Jumping ahead to 1811, under “Avogadro’s Hypothesis”:
The neglect of Avogadro’s hypothesis stopped when Stanislao Cannizzaro promoted it at an 1860 conference in Karlsruhe. It looks like Faraday was writing during the period when atomic weights hadn’t yet been sorted out.
Pingback: The Giant’s Shoulders #11 | Curving Normality
This post has been selected for Scientia Pro Publica. Please advertise the carnival on your blog and we hope to see your posts included in the future. Congratulations!
http://network.nature.com/people/primatediaries/blog/2009/05/18/scientia-pro-publica-4-in-memory-of-stephen-jay-gould
Thank you so much! This would be really helpful for my Chemistry project that is due in a few hours sadly, my procrastination is getting out of hand, but yeah, I been looking for an article about Faraday and the atomic model ALL DAY! Thank you again 😀
Glad it helped!