![]() It is fair to say that the decade surrounding the start of the 20th century was an amazingly perplexing time to be a physicist. Mounting experimental evidence strongly suggested that something was amiss with classical physics, especially in the understanding of matter on the atomic level. With the exception of a few tantalizing hints, however, almost nothing was known of atomic structure.
It is fair to say that the decade surrounding the start of the 20th century was an amazingly perplexing time to be a physicist. Mounting experimental evidence strongly suggested that something was amiss with classical physics, especially in the understanding of matter on the atomic level. With the exception of a few tantalizing hints, however, almost nothing was known of atomic structure.
The lack of information led to a flurry of speculation. In a post I wrote some time back, we discussed some eight hypothetical models of the atom that were introduced, many of them by the greatest scientists of the time. I noted that there were three important clues to atomic structure that theoreticians had to work with:
- The presence of electrons. It was well-known that atoms contained electrons.
- The periodic table. Presumably any model of atoms would have to explain the regular structure of the periodic table.
- The Balmer/Rydberg formula. Atoms were known to absorb and radiate light only at discrete frequencies. In the 1880s Balmer and Rydberg developed an empirical formula that perfectly characterized the radiative spectrum of hydrogen, and closely matched other atoms. An atomic model would need to explain this experimentally-observed interaction between light and matter.
There was at least one other very important clue to the nature of atoms that I did not mention in my earlier post: radioactivity. Since the late 1800s, physicists and chemists were aware that certain heavy atoms could give off unusual radiation of a previously unknown nature. Three types of radioactivity were observed, dubbed alpha, beta, and gamma rays. Ultimately, a comprehensive description of the atom would have to explain why some atoms were radioactive and others were not.
At least one author drew his inspiration for atomic structure directly from the observation of radioactivity. In 1903, Paris scientist Filippo Re wrote a short paper titled, “Hypothèse sur la nature des corps radioactifs,” in which he speculated that atoms coalesce from a cloud of constituent parts much like stars form from the condensation of gas in nebulae. This model provides yet another fascinating insight into the thinking of physicists of the time, and we discuss Re’s entire paper here.
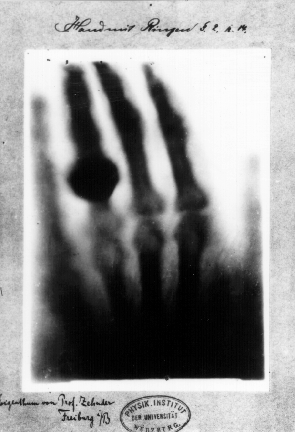
It is helpful to quickly review the history of the study of radioactivity* up to Re’s paper. We can start with the serendipitous discovery of X-rays in 1895 by Wilhelm Röntgen. He was involved in investigations of cathode rays (streams of electrons) when he noticed that a fluorescent screen was glowing, even though a cardboard obstruction stood between the cathode ray tube and the screen. Not long after the discovery, Röntgen took the first X-ray image of the human body, using his wife as a subject (source):
X-rays immediately became a scientific sensation. They captured the imagination of Henri Becquerel, a Parisian researcher in phosphorescence and fluorescence, who wondered if such “glow in the dark” materials might also give off X-rays. He wrapped photographic plates in black paper to protect them from sunlight, then placed a sample of uranium potassium sulfate on the plate in the Sun. Becquerel expected the uranium to fluoresce X-rays, which would then darken the photographic plate, and indeed he found this to be the case.
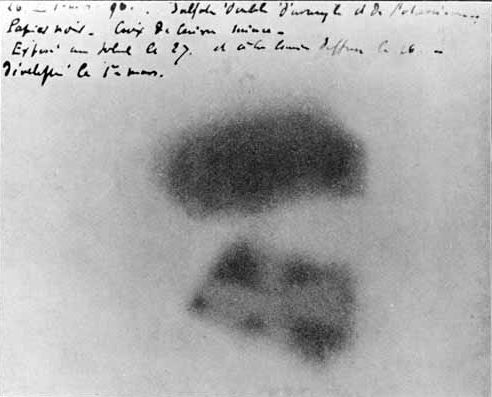
However, in a serendipitous discovery curiously parallel to the discovery of X-rays, poor weather forced Becquerel to put his experiments on hold, and he placed undeveloped plates away for a time in a drawer with the uranium still on top of it. He still developed the plates, expecting that perhaps some lingering phosphorescence might have occurred. To his surprise, the plates were even more strongly darkened than ever! Further experiments showed that the effect was not phosphorescence at all, and that uranium in any form gave off this mysterious, long-lasting radiation. Becquerel referred to this radiation as “uranic rays”; much to his chagrin others started referring to them as “Becquerel rays”. One of Becquerel’s images produced by uranium salts is shown below (source):
The dark spots are the pieces of uranium; the light cross in the lower image is from a metal Maltese cross placed between the uranium and the photographic plate, absorbing the Becquerel rays and therefore darkening the plate less.
Curiously, this potentially new form of radiation didn’t attract much attention for several years; in part this may have been due to the relatively poor image quality compared to X-rays. The attention began to shift to radioactivity, however, when the discoveries of the amazing and incomparable Marie Curie became known (source):
Remarkably, it seems that hardly anyone thought to look for radioactivity in other elements after Becquerel’s discovery. Around 1898, Curie demonstrated that thorium, one element lower on the periodic table from uranium, also possessed radioactive properties. To her dismay, she was beaten to publication of this result by two weeks by a German lecturer Gerhard Schmidt. She also noticed, however, that pitchblende, an oxide of uranium, emitted more radiation than the pure metal. She correctly deduced that another radioactive substance must be present in the pitchblende, and she and her husband Pierre crushed hundreds of kilograms by hand in order to isolate the mysterious elements. In 1898 they isolate polonium, a new element and a radioactive one. There was something even more radioactive present in even smaller quantities, and it took until 1902 to collect even a tenth of a gram of another new substance, radium.
Radium proved to be a true conundrum. It is over a million times more radioactive than uranium, and at first glance seemed to be a limitless source of energy; it is actually always warmer than the surrounding environment, which raised questions about thermodynamics as well. It and other radioactive substances turned out to be unaffected by extreme heat and cold. Perhaps most surprising, the radioactivity of one substance could induce secondary radioactivity in a nearby, inactive, substance.
Speculation about the origin and nature of radioactivity exploded. Some suggested that heavy atoms somehow absorb energy from the surrounding environment. Others wondered if the idea of conservation of energy needed to be scrapped altogether. (It wouldn’t be the last time this was suggested, either — before the discovery of the neutrino, many particle physics experiments seemed to violate conservation of energy and momentum.)
Before the mystery of radioactivity was solved, it would also be discovered that elements could transmute — turn into one another — in the process. Of the three legendary “holy grails” of ancient science — perpetual motion, the Philosopher’s Stone (lead into gold), and eternal life — radioactivity seemed to hold the key to two of them. It is perhaps no surprise that many believed that radioactivity would provide healing powers, as well; chemist Frederick Soddy, a colleague of Ernest Rutherford, even advocated in the British Medical Journal that inhaling radium could be a cure for tuberculosis!
It was into this crazed community that Filippo Re offered his theory of radioactivity. I have been unable to find much information about Re himself; he was a French scientist and evidently a colleague of Henri Becquerel, who presented Re’s paper in the French Académie des Sciences in 1903. The title of the paper may be translated as, “Hypothesis on the nature of radioactive bodies”; we take a look at it below. The translation is provided by Babelfish, with modifications on my part to clean up the clunky electronic translation.
Re begins by stating his fundamental assumption, one which had become more or less inescapable by observations of radioactivity:
Many facts result in admitting that atoms are not made of continuous matter, but of particles of a comparable nature or a different nature. The existence of radioactive bodies confirms this assumption, because one could not imagine rigid atoms giving rise to the complex phenomena of radioactivity.
Re is pointing out that radioactivity strongly suggests that atoms are made up of discrete particles, joined together; these particles may be of the same type or of a variety of types. Neither the proton nor the neutron had been discovered at this point in history; the fact that atoms could spit out pieces, however, made it clear that atoms were compound objects themselves.
That being the case, it seems natural to suppose that these particles that constitute atoms were previously free, and that they constituted an extreme nebula of tenuity; that, subsequently, they met around the centers of condensation, giving rise to suns infinitely small which, by a later process of contraction, took stable and final forms, which would be the atoms of the elements that we know and which we could compare with small extinct suns. The larger suns, which are not extinct, would constitute the atoms of the radioactive bodies.
Here we get immediately to the point! If an atom consists of a bunch of smaller particles, bound together, it is not difficult to imagine that those particles were once separated and were drawn together by some attractive force.
There is no data to back this up, so Re draws an analogy to star formation. A star generally forms when a massive interstellar cloud of dust (a nebula) collapses under its own gravity to a state of high density, initiating fusion. Re imagines an atom forms the same way, with a “nebula” of elementary particles being drawn together by unspecified forces to form the atom.
Such a process could be imagined to take time. As the pieces of the atom draw closer together, they release energy in the process. Re visualizes radioactive atoms to be atoms which have not finished this contraction process, while non-radioactive atoms have contracted to their final state and are therefore “extinct”.
This hypothesis, of which the degree of legitimacy is not less than that concerning the hypothesis of the formation of worlds, makes it possible to explain:
1: Why radioactive bodies have a very high atomic weight;
2: Why they release energy, which would be due to the contraction of their atoms.
Presumably Re imagines that the contraction process involves atoms ejecting much of their extraneous mass as radioactivity, therefore becoming lighter. This ejection carries away energy as well. This image of a radioactive atom as a body that is settling into a final state, rather than an unchanging object that is continuously giving off energy, settles the concern of energy conservation at the very least.
Moreover many of the phenomena which they present do not differ from those offered by the sun, like the rays luminous, calorific, actinic and the discharge of the electrified bodies. Concerning the induced radioactivity, several observations prove that air, rain and snow, coldly fallen, are radioactive; it does not seem improbable that their radioactivity is due to the action of solar light.
Here Re seems to be going out on a limb much further — he seems to be arguing that radioactive atoms really are miniature suns of a sort! Just like radioactive atoms can induce secondary radiation, it seems that sunlight can produce secondary radiation in air, rain and snow. “Luminous” here refers to visible light, “calorific” refers to infrared light, and “actinic” refers to ultraviolet light.
One can say nothing on the influence exerted by the magnetic fields. Indeed, it differs from that exerted on the solar rays, but one must notice that the conditions of observation are very different: in the first case, the field surrounds the radioactive body very close to him; in the second, it is well far from the source. However it is provided that the radioactive bodies must modify the magnetic state, as one observed the variation of the terrestrial magnetism caused by the sun.
This paragraph is a bit harder to understand. I assume that Re is referring to observations that magnetic fields can affect the direction of charged radioactive particles.
One could object that, if the things were thus, the radioactivity should disappear when one subjected the radioactive bodies to very low temperatures, like that from the liquid air; while it is invariable. But the objection does not have more value than that which one would do while saying than the energy released by the sun should decrease in an appreciable way and to even disappear in few years, since it is surrounded by the cold spaces sidereal. That would have come about a long time ago, if the sun were an extreme body.
The solar analogy comes to Re’s rescue here again. As we have noted, experimenters had already found that radioactivity is completely unaffected by cold temperatures. Not a problem for Re’s theory — the Sun is also unaffected by cold temperatures!
Finally the fact that minor amounts of radium release from the remarkable quantities of energy, should not astonish us. Indeed, let us reflect that, by the powerful means of disintegration which we have, we have not yet managed yet to separate the components of the atoms; one must conclude that the energy, released in their formation, must have been of an order of magnitude higher than those which one observes when the contraction is due to forces of gravitation, molecular or atomic. The atoms of the radioactive bodies, not having taken yet their final arrangement and being on the contrary in the state of formation, must thus emit great quantities of energy.
This final paragraph of Re’s paper has perhaps the most deep philosophical implications. The amount of energy released per radium atom was of a magnitude unprecedented in the scientific community. Re turns this observation on its head and points out that it must therefore take immense amounts of energy to break apart an atom, and that hypothesis agreed with the fact that nobody had yet been able to break an atom apart!
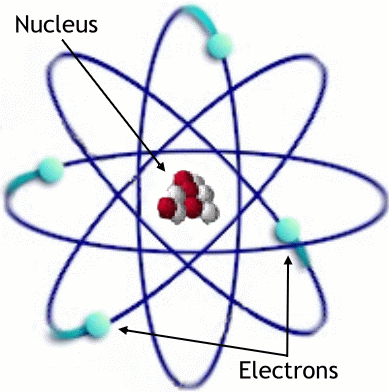
It is natural to ask, after reading Re’s paper: how much did he get right? Today, the structure of the atom is well-understood; it roughly is of a “nucleo-planetary” form (source):
An atom consists of negatively-charged electrons orbiting around a small, heavy, positively-charged nucleus. The nucleus itself consists of a collection of positively-charged protons and chargeless neutrons.
The known properties of the atom in Re’s time were a mixture of properties due to either the electrons or the nucleus. The observed electrons in the atom come (mostly) from the orbits around the nucleus. The Balmer/Rydberg formulas arise from the transition of the orbiting electrons from one orbital state to another. The periodic table, on the other hand, is dictated by how many protons are present in the nucleus, known as the atomic number. Different chemical elements are dictated by differing numbers of protons.
The nuclei of some elements are inherently unstable; they can eject energy or collections of protons and electrons to transmute into other elements. This process is what is observed as radioactivity.
Re’s specific model of the atom as a miniature condensing star is quite far from the reality. From a philosophical point of view, though, Re was correct in assuming that radioactive elements are atoms that have not yet “settled” into their lowest energy state.
It is perhaps unsurprising that Fillipo Re would choose a solar model for the atom, however; transliterated into English, “Re” is the name of the ancient Egyptian sun god (source):
In ancient Egyptian mythology, Re created the world, and the rising sun represented the symbol of creation. What better name for a man who would speculate on the fundamental building blocks of all of the world?
***********************
Update: I completely forgot to add the footnote (*) when I first wrote this post! (Probably because I finished it after midnight.) I stumbled upon a discussion of the paper by Re in the excellent book by Alex Keller, The Infancy of Atomic Physics (Dover, NY, 2006). I found Re’s paper when I, on a lark, flipped open the book to a random page and started reading. I still haven’t read Keller’s book from beginning to end, but every time I go through a chapter I find a treasure trove of fascinating information. My discussion of the history of radioactivity is roughly based on his description.
***********************
Fillipo Re (1903). Hypothése sur la nature des corps radioactifs Comptes rendus hebdomadaires des séances de l’Académie des sciences, 136, 1393-1395



You write, Becquerel referred to this radiation as “uranic rays”; much to his chagrin others started referring to them as “Becquerel rays”.
Wilhelm Röntgen went even further. In his testament he explicitly stated that he did not want his discovery to be named after himself, a request that the Germans promptly ignored and today X-rays are called Röntgenstrahlen in German. Although he won the Nobel prize for his discovery of X-rays (his own name for the phenomenon) he refused to patent the discovery saying that such discoveries belong to the general public and not the discoverer.
It’s funny to note that lots of great scientists are reluctant to have things named after them. My former thesis advisor Emil Wolf is known for the theory of “correlation-induced spectral changes”, which became known as “Wolf shifts”. He has expressed his dismay at this latter naming time and again.
X-Strahlen is too short to be a German word, anyway; at the very least, Röntgen should have added a Zug or a Schlag in there somewhere.
Hmmmm. Sounds to me like a reference to solar effects disrupting the Earth’s magnetic field. Quoth the always-infallible Wikipedia:
That’s the same FitzGerald of the Lorentz-FitzGerald contraction. (His childhood tutor was the sister of George Boole, he of Boolean logic, and FitzGerald’s maternal uncle George Johnstone Stoney coined the word electron. Small historical world!)
That is almost certainly what it refers to; the connection that I’m not sure about is what magnetic effects Re is referring to as regards the atom. It is interesting to note that the analogy between Sun and atom holds merit, though not in the way Re thought: since the Sun is powered by nuclear fusion, of course its emanations are of the same nature as that produced by individual radioactive atoms.
I have read Conquest of Cold 0 Kelvin, and was glad to see I could understand the relationship of Helium I’s transition into Helium II when the book mentioned the arising out of the Fermi surface phonons and rotons. All was clear. The entry into the world of cold was in reality a sychronized dance rule by quantized phonons. The entire book made clear in a sentence. Wonderful.
I have been looking for a book that would do such a succinct job in explaining the transition into the world of hot at the Curie point. It appears that our theories fall with each new experiment. Is there a clear transition whose rule holds?
Unfortunately, I’m not familiar with a book that talks about the transition into the ultrahot regime. 😦 Please let me know if you find one!
Filippo Re Capriata (1867-1908) was not a French. He was a professor of physics at the Messina University, born in Licata (Sicily, Italy). He died during the terrible earthquake of Messina in 1908. A correspondant of Becquerel, he made studies on radium and other newly discovered elements. In a letter to L´eclairage électrique” , a Paris scientific magazine, send in the same 1903, Re exposed a curious system to transmit images using the properties of selenium, thus somehow anticipating television.
Thanks for the correction! As I noted in the post, there isn’t a lot of information about him online. I’m guessing I couldn’t find info because in the paper his name is only listed as Filippo Re. Do you have some insight into why the Capriata would have been left off the publication?
hmm