![]() While researching a recent post on the history of nuclear physics (here), I happened across a short but rather fascinating letter written in 1903. It seems to be the first article in print that makes the connection between the processes which cause radioactivity and the Sun’s radiation! Though this article, by W.E. Wilson, seems to have been mostly forgotten*, it provides yet more insight into the steady yet stumbling process by which scientists come to understand complicated and mysterious phenomenon.
While researching a recent post on the history of nuclear physics (here), I happened across a short but rather fascinating letter written in 1903. It seems to be the first article in print that makes the connection between the processes which cause radioactivity and the Sun’s radiation! Though this article, by W.E. Wilson, seems to have been mostly forgotten*, it provides yet more insight into the steady yet stumbling process by which scientists come to understand complicated and mysterious phenomenon.
A little background will be helpful here. The phenomenon of radioactivity was first discovered** serendipitously in 1896 by Henri Bequerel, who noted that photographic plates were darkened in the presence of uranium. Evidently uranium spontaneously emitted a new form of radiation distinct from X-rays and other waves in the electromagnetic spectrum.
Bequerel’s first observation didn’t draw that much attention, as it was overshadowed by the remarkable promise of X-rays, discovered around the same time. However, Marie Curie began to investigate whether other elements also produced radioactivity, and in 1898 she demonstrated that thorium was also radioactive. Perhaps more significantly, she noticed that different ores of uranium seemed to possess different degrees of radioactivity, and led her to speculate that a new radioactive element was hidden in small quantities in certain uranium composites. By 1902, the Curies had collected a small quantity of this new element, dubbed radium.
Radium really caught people’s attention. It is roughly a million times more radioactive than uranium, and to researchers at the time it appeared to be a nearly limitless source of energy. A sample of radium is actually warmer than the surrounding environment, making it appear to violate well-established thermodynamic principles as well.
The prodigious energy output of radium made it a natural candidate to explain one of the other great mysteries of science: the source of the Sun’s energy. This mystery had occupied some of the great minds of the 19th century, and no satisfactory answer had yet been achieved. In 1862, Lord Kelvin suggested that the Sun is a molten body that radiates heat as it cools, possibly gaining more heat through the impact of meteorites; from this model he estimated the age of the Sun as likely being 50 million to 100 million years. A similar model for the Earth suggested it to be between 20 million and 400 million years.
These results were on the surface somewhat paradoxical (which came first, the Earth or the Sun?) and also at odds with evolutionary estimates of the Earth’s age, which required billions of years. Kelvin’s model, however, was one of the biggest arguments against Darwin’s evolutionary theory at first, and Kelvin one of the biggest opponents of the model. Darwin in fact found Kelvin’s estimate daunting, and referred to Kelvin coming along “like an odious spectre” to thwart his reliance on long evolutionary epochs.
The mysterious new radioactive power of radium pretty much cried out as an answer to the mystery of the Sun’s energy. It is perhaps not surprising that in 1903, only a year after the discovery of radium, W.E. Wilson suggested exactly this.
William Edward Wilson (1851-1908) was an Irish astronomer with no university education; due to a delicate constitution, he was home schooled. Under the influence of his father, a man of intellectual leanings, Wilson developed a passion for astronomy and purchased a 12 inch reflecting telescope in 1871, setting it up in a dome in the garden of the family home at Daramona House, in County Westmeath.
The Daramona Observatory, c. 1900. (From EAAS website)
From the Daramona Observatory, Wilson made a variety of astronomical investigations, but his main focus was on solar observations. Among his achievements (with colleagues) were an estimate of the Sun’s temperature (strikingly close to the modern value) and the first cinematograph (movie) of sunspot activity. In addition to his investigations of the Sun, Wilson also dabbled in X-ray studies, which probably made him acutely aware of developments in radioactivity, as well.
W.E. Wilson, c. 1900 (also from EAAS website).
Wilson wrote a letter to Nature that was printed in the July 9, 1903 issue, vol. 68, p. 222. The text of this letter, in its entirety, is below:
Radium and Solar Energy
The extraordinary discovery that radium has the property of continuously radiating heat without itself cooling down to the temperature of surrounding objects may possibly afford a clue to the source of energy in the sun and stars.
Taking the Curies’ observation that one gram of radium can supply 100 calories per hour, I thought it would be of interest to compute how much radium would suffice to supply the sun’s output of energy.
Taking from Langley’s observations that this is equal to 828,000,000 calories per square cm. per hour, I find that 3.6 grams of radium per cubic metre of the sun’s volume would supply the entire output.
It may be possible that at solar temperature radium is capable of much more energetic radiation, and, if so, the 3.6 grams might be reduced to a much smaller figure.
Daramona, July 1 W.E. Wilson
Using simple back of the envelope calculations, Wilson suggests that a relatively tiny amount of radium (3.6 grams per cubic meter of the Sun’s volume) could provide all of the observed energy of the Sun’s output!
Wilson’s letter provides a great lesson as to how science works: in its particulars, it is in fact entirely incorrect, but nevertheless it was at the time a dramatic step in the direction of the “right” answer.
Today we know that the radioactivity of radium and the radiation emitted by the sun are, in essence, two aspects of interactions that can occur with the nuclei of atoms. The radioactivity of radium is the result of the process of nuclear fission, in which an atomic nucleus breaks into two or more pieces; the radiation of the sun is the result of the process of nuclear fusion, in which two or more atomic nuclei are fused together into one. For those who may not remember exactly what these processes entail, a brief review is in order.
An atom consists of a very tiny nucleus of positive electric charge “orbited” by negatively charged electrons. An electrically neutral atom possesses an equal amount of positive and negative charge:
The nucleus itself always has a discrete number of positive electric charges within it; this number, called the atomic number, determines the chemical properties of the atom and its place on the periodic table. All of traditional chemistry results from the interactions of electric charges between different atoms.
The periodic table of elements. The atomic number, the top number in the box, indicates the amount of positive charge in the nucleus. (From Brewton-Parker College.)
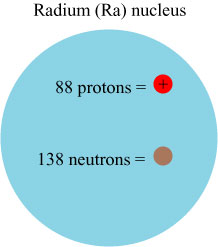
The nucleus of an atom itself consists of pieces, namely a collection of positively-charged protons and uncharged neutrons. The number of protons is the atomic number, and therefore characterizes the element. The total number of protons and neutrons in an element is called the atomic mass number, and different isotopes of an element are labeled by this mass number. For instance, the most common form of radium has an atomic number of 88, and an atomic mass number of 226, and is abbreviated 226Ra; it therefore has 138 neutrons. Schematically, we might sketch the nucleus as:
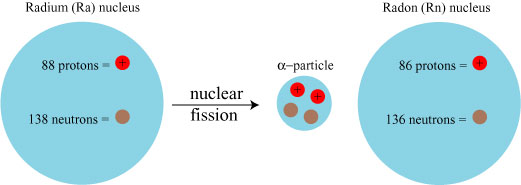
Heavier elements (those with higher atomic number) tend to be unstable; that is, the energy of the nucleus as a whole is larger than the energy of the nucleus in pieces. Such a nucleus will tend to break apart by nuclear fission; in the case of radium, it releases an alpha particle, which consists of two protons + two neutrons, at high speed. What remains is an atom with a smaller atomic mass number, namely 222Rn, Radon 222:
Lighter elements (those with smaller atomic number) tend to be stable; that is, the energy of the nucleus as a whole is smaller than the energy of the energy of the pieces. If by some means we bring these pieces close enough together, a larger nucleus will form, and energy will be released; this is the process of nuclear fusion.
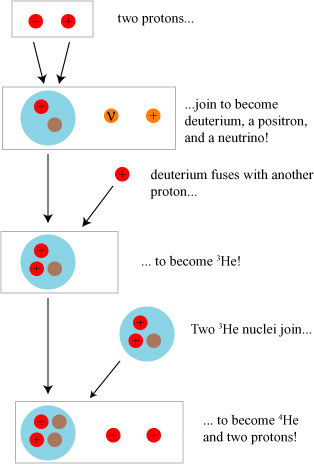
In the Sun, the active process is what is generally referred to as the proton-proton chain reaction, in which multiple hydrogen nuclei (1H) come together in a complicated process, the outcome of which is a stable nucleus of helium (4He). One possible means by which this occurs is illustrated below:
The subtle part of this process is in fact the first step, which involves the so-called weak nuclear force. In the weak process depicted here, a proton becomes a neutron, a positron (anti-electron), and an electron neutrino.
Nuclear fusion in general requires a high density of nuclei at high temperature; the nuclei must be packed together tight enough to overcome the natural repulsion of their positive electric charges. In the Sun, the crushing force of gravity performs the process. Here on Earth, a number of national labs are attempting to produce the same result using high-power lasers to smash atoms together; in Rochester, NY, the Laboratory for Laser Energetics has the massive Omega laser system attempting to create laser-induced fusion***.
Both nuclear fusion and nuclear fission arise from fundamental forces of nature that were unknown in Wilson’s time, the creatively-named strong nuclear force and weak nuclear force. The strong nuclear force is what holds protons and neutrons together, while the weak force is involved in a number of other radioactive decay processes. The nucleus itself would not be unveiled until 1911, after Ernest Rutherford interpreted his famous gold foil experiment as indicating the presence of a dense, central atomic core.
So Wilson was completely incorrect in the details of his letter — the Sun is powered by nuclear fusion, not the nuclear fission of radium — but he was correct in realizing that the prodigious energy output of the Sun was somehow connected to this newly discovered phenomenon of radioactivity. Simply the realization that new physics was required to explain the Sun’s output was a tremendous step in the right direction.
Wilson’s suggestion seems to have been overshadowed by that of a much more prominent scientist, namely Ernest Rutherford. In Spring of 1904, while giving a lecture on radium at which Lord Kelvin was also in attendance, Rutherford argued that the presence of radium in the Earth would provide an internal source of heat, destroying the assumptions used in Kelvin’s estimate of the age of the Earth. By extension, it was not difficult to see that the same source of heat could power the Sun, thus punching a hole in Kelvin’s other estimate.
A true understanding of the nature of the Sun’s power would take many more years to develop. Around 1920, astrophysicist Arthur Eddington (1882-1944) would be the first to argue that nuclear fusion plays a fundamental role in stellar objects. It was not until the 1930s, however, that astrophysicists Subrahmanyan Chandrasekhar (1910-1995) and Hans Bethe (1906-2005) detailed the specific nuclear processes described above.
Wilson’s early speculation seems to have been more or less forgotten by history. This is perhaps not unfair, as it was a small contribution in the overall understanding of the Sun and nuclear fusion. Wilson did not suffer, anyway; in 1896 he was elected a Fellow of the Royal Society, and in 1901 the home-educated scientist received the degree of Doctor of Science from the University of Dublin!
************************************
* I came across a reference to this letter in Alex Keller’s excellent history, The Infancy of Atomic Physics, which has provided fodder for at least three blog posts now. The book was written in 1983, clearly pre-Google: Keller refers to “W.E. Wilson of Daramona (wherever that might be)”. I found a wealth of info on Daramona with a simple search!
** It is a bit inaccurate to refer to Bequerel as having “first discovered” radioactivity, as apparently others had observed the phenomenon years earlier. These earlier researchers, however, failed to note the significance of the effect, and didn’t perform a systematic study as Bequerel would.
*** As a graduate student, I used to live around the corner from the LLE. I got a kick out of making my parents nervous by telling them that I had a nuclear fusion reactor as a neighbor.
************************************
WILSON, W. (1903). Radium and Solar Energy Nature, 68 (1758), 222-222 DOI: 10.1038/068222a0






Whoops — big mistake in my discussion of nuclear fission! Radium decays into radon, not polonium. I’ll fix the figure when I get back from work this evening.
Update: Fixed!
An interesting tie in to this whole story is that one of the first people to publish on the idea that radioactivity might be the key to the age of the sun was George H. Darwin in Nature in 1903 http://www.nature.com/nature/journal/v68/n1769/pdf/068496b0.pdf
Ah, you scooped me — I was thinking of writing a post about George’s role sometime in the future! 🙂
Pingback: positive atoms | led
This was interesting and helpful to me in my research, thans!