“My name is Erasto B Mpemba, and I am going to tell you about my discovery, which was due to misusing a refrigerator.”
With those words, Tanzanian student Erasto Mpemba entered scientific history, and also sparked a scientific mystery and controversy that remains ongoing today, some 40 years later!
The phenomenon Mpemba found is now known as the Mpemba effect, and is the very counterintuitive idea that, under certain circumstances, a quantity of very hot/boiling liquid can freeze faster than an equal quantity of cold liquid!
![]() How is this possible? The remarkable thing is that nobody really knows, even though the first observations were reported to the scientific community in 1969. The story of the discovery, and the consequent mystery, is worth a bit of exploration — and the Mpemba effect carries numerous important lessons about the nature and method of scientific discovery.
How is this possible? The remarkable thing is that nobody really knows, even though the first observations were reported to the scientific community in 1969. The story of the discovery, and the consequent mystery, is worth a bit of exploration — and the Mpemba effect carries numerous important lessons about the nature and method of scientific discovery.
Mpemba made his accidental discovery in Tanzania in 1963, when he was only 13 years old and in secondary school. In spite of widespread disdain from his classmates, he surreptitiously continued experiments on the phenomenon until he had the good fortune in high school to interact with Professor Denis Osborne of the University College Dar es Salaam. Osborne was intrigued, carried out his own experiments, and in 1969 the two published a paper in the journal Physics Education.
This article is, in my opinion, one of the most remarkable in all of the history of physics. Aside from its title, “Cool?”, it is also unusual in being presented in two parts: Mpemba gives a first person account in his own words of his discovery in the first half, and Osborne picks up the story and describes the follow-up experiments in the second half. Mpemba’s own account is so charming and fascinating that it is worth quoting from liberally:
My name is Erasto B Mpemba, and I am going to tell you about my discovery, which was due to misusing a refrigerator. All of you know that it is advisable not to put hot things in a refrigerator, for you somehow shock it; and it will not last long.
In 1963, when I was in form 3 in Magamba Secondary School, Tanzania, I used to make ice-cream. The boys at the school do this by boiling milk, mixing it with sugar and putting it into the freezing chamber in the refrigerator, after it has first cooled nearly to room temperature. A lot of boys make it and there is a rush to get space in the refrigerator.
One day after buying milk from the local women, I started boiling it. Another boy, who had bought some milk for making ice-cream, ran to the refrigerator when he saw me boiling up milk and quickly mixed his milk with sugar and poured it into the icetray without boiling it; so that he may not miss his chance. Knowing that if I waited for the boiled milk to cool before placing it in the refrigerator I would lose the last available ice-tray, I decided to risk ruin to the refrigerator on that day by putting hot milk into it. The other boy and I went back an hour and a half later and found that my tray of milk had frozen into ice-cream while his was still only a thick liquid, not yet frozen.
I asked my physics teacher why it happened like that, with the milk that was hot freezing first, and the answer he gave me was that “You were confused, that cannot happen”. Then I believed his answer.
Here we have the beginnings of a classic story of science — an accidental discovery, scoffed at by the “establishment scientists”.
Mpemba might have given up at that point, but he encountered a friend who sold ice cream for a living, and that friend happened to mention that many vendors would use boiling water to make their ice cream! It was already common knowledge amongst them, apparently, that a boiling mixture could freeze quicker.
After passing my O level examination, I was chosen to go to Mkwawa High School in Iringa. The first topics we dealt with were on heat. One day as our teacher taught us about Newton’s law of cooling, I asked him the question, “Please, sir, why is it that when you put both hot milk and cold milk into a refrigerator at the same time, the hot milk freezes first?” The teacher replied: “I do not think so, Mpemba.” I continued: “It is true, sir, I have done it myself” and he said: “The answer I can give is that you were confused.” I kept on arguing, and the final answer he gave me was that: “Well, all I can say is that that is Mpemba’s physics and not the universal physics.” From then onwards if I failed in a problem by making a mistake in looking up the logarithms this teacher used to say: “That is Mpemba’s mathematics.”
And the whole class adopted this, and anytime I did something wrong they used to say to me “That is Mpemba’s…”, whatever the thing was.
Here the high school teacher failed miserably — ridiculing a student is pretty much the worst thing one can do in a science classroom! Fortunately, Mpemba was not deterred:
Then one afternoon found the biology laboratory open, and there was no teacher. I took two 50 cm³ beakers, one I filled with cold water from the tap and the other with hot water from a boiler and quickly put them in the freezing chamber of the laboratory refrigerator. After one hour I came back to look and I found that not all the water had been changed into ice, but that there was more ice in the beaker which had hot water to start with than in the one which had cold water. This was not really conclusive. So, I planned to try it again when I had the chance.
Before he had this chance, however, Professor Osborne came to lecture on physics, giving Mpemba a valuable opportunity:
When Dr Osborne visited our school we were allowed to ask him some questions, mainly in physics. I asked: “If you take two similar containers with equal volumes Of water, one at 35 °C and the other at 100 °C, and put them into a refrigerator, the one that started at 100 °C freezes first. Why?” He first smiled and asked me to repeat the question. After I repeated it he said: “Is it true, have you done it?” I said: “Yes.” Then he said: “I do not know, but I promise to try this experiment when I am back in Dar es Salaam.” Next day my classmates in form six were saying to me that I had shamed them by asking that question and that my aim was to ask a question which Dr Osborne would not be able to answer. Some said to me: “But Mpemba did you understand your chapter on Newton’s law of cooling?” I told them: “Theory differs from practical.” Some said : “We do not wonder, for that was Mpemba’s physics.”
There are many remarkable points in this short passage. First of all, we see an admirable open-mindedness of Professor Osborne in his dealings with Mpemba, and that open-mindedness would quickly benefit them both. Conversely, we see a dangerous “groupthink” amongst Mpemba’s classmates regarding science, in which they are genuinely offended by Mpemba questioning the status quo. Mpemba shows great wisdom in his answer: “Theory differs from practical”. This is an important point for anyone studying physics: we like to create simplified models to explain nature, but those models often lose real-world aspects in the process of stripping them down.
Mpemba actually continued his experiments in a kitchen refrigerator, with the permission of kitchen staff, and convinced his classmates and the headmaster of his school of the accuracy of his findings.
At Dar es Salaam, Osborne was true to his word and looked into the phenomenon himself. As he notes in the continuation of the paper,
It seemed an unlikely happening, but the student insisted that he was sure of the facts. I confess that I thought he was mistaken but fortunately remembered the need to encourage students to develop questioning and critical attitudes. No question should be ridiculed. In this case there was an added reason for caution, for everyday events are seldom as simple as they seem and it is dangerous to pass a superficial judgment on what can and cannot be. I said that the facts as they were given surprised me because they appeared to contradict the physics I know. But I added that it was possible that the rate of cooling might be affected by some factor I had not considered.
Osborne sets a great example for all physics educators! It can be difficult at times, but “No question should be ridiculed” would be a great part of a “Hippocratic oath” for teachers.
One other anecdote from Osborne’s account is worth quotation:
At the University College in Dar es Salaam I asked a young technician to test the facts. The technician reported that the water that started hot did indeed freeze first and added in a moment of unscientific enthusiasm: “But we’ll keep on repeating the experiment until we get the right result.”
I leave it as an exercise to the reader to explain what is scientifically wrong with the technician’s attitude!
So what did Osborne’s research show? He placed a 100 cm³ beaker filled with 70 cm³ of water on a sheet of insulating foam in a freezer, and timed how long it took for the water to freeze. For temperatures up to 20 °C, the time was roughly proportional to the temperature above freezing, up to a maximum of 100 minutes at 20 °C. For higher temperatures, however, the time dropped dramatically, down to 40 minutes for 80 °C water!
Later experiments on the effect have been far less conclusive than Mpemba and Osborne’s. Some have seen similar results, while others have observed no effect at all! The appearance of the Mpemba effect apparently depends very strongly on the specific experimental circumstances, and is much harder to reproduce than the original paper would imply. A number of physicists seem skeptical that the effect truly exists at all!
With this in mind, however, it is worth noting that Mpemba’s observation was in fact noted earlier by others over the course of the past two thousand years. Around 350 B.C.E., Aristotle, in giving an explanation of hail in his book “Meteorology”¹, noted:
The fact that the water has previously been warmed contributes to its freezing quickly; for so it cools sooner. (Hence many people, when they want to cool water quickly, begin by putting it in the sun. So the inhabitants of Pontus when they encamp on the ice to fish (they cut a hole in the ice and then fish) pour warm water round their rods that it may freeze the quicker; for they use the ice like lead to fix the rods.) Now it is in hot countries and seasons that the water which forms soon grows warm.
Others who argued for the existence of a Mpemba effect, under some circumstances, include Roger Bacon in the 13th century, and Francis Bacon and René Descartes in the 17th century. It was Mpemba and Osborne, however, who brought it to the attention of modern science.
Before delving into some of the explanations for the effect, it is important to explain why the natural objection to its existence is not necessarily applicable. A natural argument is as follows. “Suppose we have two equal glasses of water, one at 100 °C and one at 30 °C. In order for the water at 100 °C to freeze, it must first cool to 30 °C. The time it takes to freeze is therefore the amount of time it takes to go from 100 to 30 plus the time it takes to go from 30 to zero. It therefore must take longer to freeze than the 30 °C water.”
The flaw in this argument is assuming that a body of water is only characterized by a single parameter: its temperature. In general, there are other characteristics of the water that could be changed due to its heating; for instance, the amount of gas in solution, the presence of other solutes, the presence of convection currents, gradients of the distribution of temperature in the container. Any, all, or none of these may in fact be the culprit, but it is important to realize that a body of water not in thermal equilibrium may have its behavior characterized by a number of different properties.
A number of hypotheses have been proposed as explanations, some based on the factors mentioned:
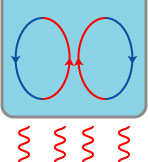
1. Convective heat transfer. When a liquid is heated, it can form convection currents that rapidly bring the hot liquid to the surface, where the heat is lost by evaporation:
Osborne noted that this convection will keep the top of the liquid hotter than the bottom, even when the temperature matches an initially cold liquid that doesn’t possess this convection cooling. This results in a faster rate of cooling that could, under the right circumstances, result in Mpemba’s observation.
2. Evaporation. A boiling or very hot liquid will lose some of its mass due to evaporation. With a lower mass, it will cool faster, possibly giving a “boost” to the Mpemba effect. Osborne already noted that evaporation alone could not account for all of the rate of cooling of the hot liquid.
3. Degassing. In a series of experiments in 1988, a Polish research group² reproduced the Mpemba effect and noted that the effect depended strongly on the amount of gas dissolved in the water. When the water was purged of air and carbon dioxide, the time to freeze became proportional to the starting temperature. The researchers suggested that the presence of gas was substantially slowing the rate of cooling, and that the heated water was purged of it.
4. Supercooling. In 1995, German scientist David Auerbach proposed³ that the Mpemba effect could be explained by supercooling, and performed experiments to back up the assertion.
Liquids begin to crystallize into a solid at the freezing point with the help of impurities around which the crystals can nucleate. In the absence of such impurities, the liquid can be cooled below its normal freezing point while remaining a liquid — it is “supercooled”. Auerbach suggests that the cold water will supercool to a lower temperature than the hot water, thus giving the hot water an “edge”. However, the reason that hot water has a higher supercooling temperature is unclear, and possibly related to the earlier noted effects.
5. Distribution of solutes. In 2009, an interesting explanation was proposed4 by J.I. Katz, who suggested that solutes present in the cold water slow the freezing process, as suggested earlier, but also that those solutes get driven from the freezing water into the as-yet unfrozen water, slowing the process further.
Other suggestions have been made, and other experiments have been done5, though no consensus seems to have been reached regarding the origins and generality of the effect. The confusion about the Mpemba effect is curiously reminiscent of the controversy around the “Archimedes death ray” controversy we have discussed earlier — the answer depends on the question being asked. Does hot water always freeze faster than cold? Almost certainly not! Are there some circumstances under which hot water freezes faster than cold? It seems likely, though nobody is sure exactly what those circumstances are. Does a negative result disprove the Mpemba effect, or was the experiment just done under the wrong circumstances? Nobody knows for certain.
The controversy no longer is of concern to Erasto Mpemba, however, who did not become a physicist himself but ended up studying at the College of African Wildlife Management. With his education, he eventually became the Principal Game Officer for the Tanzanian Ministry of Natural Resources and Tourism, working on wildlife management and conservation.
Erasto Mpemba, circa 1997 (source).
Regardless of the mystery, Mpemba’s discovery is a wonderful illustration of many important lessons in science: the significance of experiment over theory, the danger of clinging to preconceived notions, the difficulty in evaluating even seemingly simple real-world physics problems, and the importance of perseverance in the face of unreasoning denial. Furthermore, as far as I know the Mpemba effect is one of the only (if not the only) physical phenomena named after an African researcher! It is a wonderful and frustrating hint of how much intellectual potential lies untapped on the continent.
So what do I think of all the controversy? I’m not quite sure what to think! I’d love to test the Mpemba effect myself at home, but our freezer is packed to capacity6! Any enterprising experimenters out there want to give it a try? Describe your results in the comments!
********************
(Thanks to Blake Stacey for providing one of the references used to write this post!)
¹ Aristotle, “Meteorology”, in The Complete Works of Aristotle (The revised Oxford translation), (J. Barnes, ed., Princeton University Press, 1984).
² Wojciechowski, Owczarek and Bednarz, “Freezing of aqueous solutions containing gases,” Cryst. Res. Technol. 23 (1988), 843-848.
³ D. Auerbach, “Supercooling and the Mpemba effect: When hot water freezes quicker than cold,” Am. J. Phys. 63 (1995), 882-885.
4 J.I. Katz, “When hot water freezes before cold,” Am. J. Phys. 77 (2009), 27-29.
5 Esposito, De Risi and Somma, “Mpemba effect and phase transitions in the adiabatic cooling of water before freezing,” Physica A 387 (2008), 757-763.
6 This is a clear indication of my current marital status. Back in my single days, the only thing you would find in my freezer would have been a pair of frozen pizzas!
E.B. Mpemba, & D.G. Osborne (1969). Cool? Physics Education, 4, 172-175



The technician’s attitude is basically one of only wanting to find the desired result (i.e., the one in agreement with theory). The way we want Nature to work, and what it does are two very different things. Sometimes our common sense and intuition can prevail (e.g., much of physics up to about the 1830’s).
Did the experiments with three independent samples of water. Samples were 1) Tap 2) Pur filtered and 3) Fiji bottled water. All samples were 355 ml and were placed in a freezer at -12.7 C. The container was a smooth clear glass. The freezer temperature was monitored ever 30 minutes to ensure consistency. One sample was kept at room temperature 23 C while the other was heated to 90 C. The samples were checked after one hour. The heated tap water sample appeared to have developed greater nucleation that the water at room temperature. The Pur filtered water gave similar results. For the Fiji bottled water, the room temperature sample froze 79 minutes faster than the heated sample.
Very nicely done, and fascinating! It kind of makes sense that the tap water would exhibit a stronger effect, and intriguing that the filtered water did the same.
Yea, water filters are more marketing and less science, big surprise.
I’ll keep it simple and accept impurities (solute) in the water as the culprit…for now anyway!
No need to accept anything — it’s still speculation until someone really conclusively demonstrates it!
We perhaps need a different control. How about this:
Take two water samples. Heat them both to the same temperature. Then cool one of them for a set time while the other keeps the original temperature. (Or perhaps cool one of them in a freezer while the other cools at room temperature.) Measure the temperature of both after that time; one should be warmer than the other. Then freeze both of them and see which freezes faster.
If the warmer one freezes faster under these circumstances, then the difference is not in what happened before or while they were originally heated.
Heat them to the same temperature, then cool one of them to a lower temperature. Put both in closed containers half full of air (check air temperature and barometric pressure if you want to get fancy). Shake them vigorously for the same time. Then cool them. That tells you how much difference shaking in air makes. Try shaking them before you cool one, or shake them after you cool one.
I’d do it but my freezer is full too.
Nice suggestions! It does seem like a more systematic method is needed to eliminate different factors. The catch, however, is that more than one factor may contribute to the effect!
Pingback: The Mpemba Effect « adafruit industries blog
Osborne sets a great example for all physics educators! It can be difficult at times, but “No question should be ridiculed” would be a great part of a “Hippocratic oath” for teachers.
So, I’m going to be fully open minded and question even that 🙂
The problem with this is that various forms of creationists (and promoters of other kinds of pseudoscience) have done a great job of coming up with “stump the evolutionist” questions. It makes it very tricky to deal with those kinds of questions without making it look like you’re supporting the potential viability of something that’s well-documented as false. It’s tricky.
Let me add that ridicule is almost never the right way to proceed. (I don’t want to fully rule it out; bug me if you want specifics.) However, accepting all questions as valid questions does potentially open up several societal Pandora’s boxes– not hypothetical ones, but real ones that are causing problems for science and for our society in general.
“making it look like you’re supporting the potential viability of something that’s well-documented as false.”
That’s exactly what the teacher was afraid of and why he ridiculed the student. And that’s why anything that contradicts established thought (or what is thought of as “fact” like evolution) will always be fought against by the established “scientists”. Established scientists use the term “pseudo-science” much in the same way that the teacher used the term “mpemba physics”: they use it to rule out ideas they dont want or are unable to discuss openly.
Too true! Like any very broadly applied rule of conduct, there are always exceptions. One such exception would be questions that are clearly asked insincerely, such as those creationists usually ask. OTOH, if a young person was sincerely asking for an explanation of some creationist garbage they had heard/read, treating the questioner with respect is probably the best approach.
Of course, the trick is determining the difference between a sincere and insincere question! I have that problem on the blog on semi-regular occasions, where a seemingly innocent question turns out to be the opening volley of someone with an ideological axe to grind.
This difference here though is that Mpemba’s theory was testable, but up until Osborne heard it, no one bothered to test the theory, they just assumed it was wrong. Pseudoscience tends to stick around because it’s not easily testable or is based on a premise that is by its very nature is untestable.
Indeed! In fact, some serious thought has been given as to whether the Mpemba effect is an untestable or, more specifically unfalsifiable experiment. A 2006 paper by Jeng, cited below, specifically addresses this question. In short, the Mpemba effect is testable, but one has to restrict the question to a manageable set of experimental parameters: starting temperatures, container size/shape, and so on.
M. Jeng, “The Mpemba effect: When can hot water freeze faster than cold?” Am. J. Phys. 74 (2006), 514-522.
Pingback: Mpemba's baffling discovery: can hot water freeze before cold … | My Blog
As baffling as hot water freezing first is it is no wonder that there is some much controversy over global warning theory.
There’s truth to that! Thermodynamic problems, including climate issues, are some of the most difficult to cope with.
Is the effect only ever seen with water?
I’m not sure that it has been observed for any other non-water-based liquid, but evidently works for very different mixtures: Mpemba, for instance, first observed the effect with boiling milk.
When I was in physics and I proposed this idea as I heard if from a friends father. We looked it up, got all the data and attempted to test it ourselves. What we did was take three ice trays that were exactly the same. We then took water and measured it out into three beakers at exactly the same amounts. One was at room temperature, one was cooled inside a fridge, and one was heated to almost boiling.
What we found was that about half the time, the boiling water would freeze faster than the room temp water but the cooled water always froze first. We went over different theories and ideas and we tested this about 50+ times in the span of 6 weeks. The results were intermittent so our data didn’t hold any one theory or idea to be true. I can see why so many still debate over this.
Very nicely done! I’m not surprised that you found the results only work about “half the time”. One thing I didn’t mention in my post is that the later “supercooling” paper found it was a somewhat random process — the hot water was more likely to freeze first, but not with certainty. Presumably there are factors that could increase that likelihood further, but as noted nobody knows what those factors might be!
It would be good to know if these experiments are taking place in the same freezer or if the boiling water is in a different freezer than the room temperature water. That would make a big difference. If they are in the same freezer at the same time wouldn’t the cold air molecules move in favor of the hot tray because the hot air surrounding it would dissipate quicker leaving room for the slower moving cold air molecules to pile up?
I think in the original Mpemba paper the two samples were in the same freezer. It is an interesting point that merely the presence of the two together might somehow influence the results, though.
Pingback: The Mpemba Effect: A Good Case For Citizen Science? – Global Spin
hey that’s an amazing discovery by mpemba . I am going to try this experiment to make ice cream. I also thought a liquid at normal temperature would freeze first but that’s an amazing experiment. Great post looking forward to more from you!
Feel free to let us know the results of your tests in the comments! 🙂
Gosh. I haven’t ever thought about it but it simply makes no sense. I have to try the experiments. Isn’t it interesting that a simple scientific observation makes such interesting posts. Thanks son very much for posting.
If you try it, please come back and let us know what you find in the comments! And you’re welcome!
Pingback: … for that was Mpemba’s physics | schegge
Pingback: Theory Vs Practical | T a i j i K i n e s i s ~ R e d u x
To my astonishment some years ago I noticed that the pipes for distribution of water to different rooms in my house – none were covered with insulation EXCEPT the pipes for the warm water. All distribution pipes are located in the space between the ground and the house itself surrounded by the walls of the house. Since here in Norway were I live it can be more than – 30 C there always excists a possibillity of freezing of the water in the the pipes.
I asked the plummer if this was a mistake, but he answered that they only put insulation on the pipes for the warm water – and added – the pipes for the cold water ususally did not need insulation!
Thanks for the comment! The hot pipe/cold pipe phenomenon is addressed in a number of the papers of the Mpemba effect, though there seemed to be some disagreement as to whether it should be considered the same effect. It certainly is as counterintuitive!
Hot water pipes freeze first due to pressure. Hot water has expanded. So as it cools there is space in the closed pipe for the water to expand as it freezes. Cold water pipes are closer to maximum capacity at lower temps so as the ice crystals start to form there is less room for expansion. Lower temp is required to freeze as the pressure in creases.
I’m not at all convinced that under conditions where the only difference between the two samples is the temperature, and therefore the volumes, the “Mpemba Effect” holds any place in reality. The water for both sample should be boiled for a time to allow any aromatic compounds and gases to evaporate, then the common source should be allowed to cool, say to room temperature, and then a sample of a given mass should be set aside. The remainer of the source them be reheated to an arbitrary temperature, and then a second sample of equal mass be put in the same type of vessel as the first. Both should be put into the same freezer, but shielding installed to guard against radiant energy affecting the cooler sample. Alternately, the two samples could be cooled in separate freezers, although neither should have their function regulated by thermostatic means—the hotter of the two could trigger its freezer to work harder to cool it. Even if the heated water were to “catch up” to the cool water, they then would be on equal standing and would freeze at the same rate from that point.
I believe The Mpemba Effect should be defined as “the ability to propagate belief in the impossible through the use of jargon and third-hand testimonials of fictitous observed phenomenon”.
Whoa, dude. Emotion and rhetoric are not science. Its healthy to say ‘there’s something here we don’t understand. Yet.’ It’s not healthy to reject and ridicule observations, then suggest possible explanations, and propose, but not do, experiments. Or is this intentional irony?
We all make mistakes, but “third-hand testimonials of fictitous observed phenomenon” is not an honest description of 4 citations in 3 different journals, Or the original report. Are E.B. Mpemba, & D.G. Osborne liars? Are the rest somehow deluded?
The opening challenge, “I’m not… convinced that… the “Mpemba Effect” holds any place in reality.” is good theater but not-great science, particularly when, conditioned by “under conditions where the only difference between the two samples is the temperature, and therefore the volumes”.
Edited down to content, you seem to be saying,
“What happens under conditions where the only difference between the two samples is the temperature, and therefore the volume[s]?
Both sample[s] should be boiled for a time to allow any aromatic compounds and gases to evaporate, then the common source should be allowed to cool, say to room temperature, and then a sample of a given mass should be set aside.
The remainer of the source them be reheated to an arbitrary temperature, and then a second sample of equal mass be put in the same type of vessel as the first.”
I’m not sure I follow, “Both should be put into the same freezer, but shielding installed to guard against radiant energy affecting the cooler sample. ” – the warmer sample will emit heat that prevents to cooler one from freezing, while freezing itself? Doesn’t the description of the original reports suggest some experiments were done side by side, others by measuring time-to-freeze?
I think “Alternately, the two samples could be cooled in separate freezers, although neither should have their function regulated by thermostatic means—the hotter of the two could trigger its freezer to work harder to cool it.” contains a valuable suggestion- a near-boiling sample may coerce to freezer to apply more effort toward cooling. So compare two samples side by side vs one sample at a time, noting if or how long the compressor of the freezer runs. It might be worth comparing with a very large block of ice and an insulated container or working outdoors in sub-freezing weather, too.
Temperature over time should be recorded and an experiment should check for the heated water to “catch up” to the cool water, … then … would freeze at the same rate from that point.”
Bill
Very well put! And I did not include all of the published observations, either!
I’m not sure you understood the point of the theoretical explanations that were proposed. If the only real difference between the samples was the temperature, the Mpemba effect could not be possible! Every experimenter has appreciated this, and that is why the nature of the solutes has been given great attention. Your proposed experiments seem to be, in short: “If I take away anything that could cause a Mpemba effect, I should see no Mpemba effect.” That doesn’t mean it isn’t worth doing, but it kind of misses the point.
What I said about “third-hand testimonials…” was rhetorical but some lay people hear about the Mpemba Effect and think that the laws of phyics dictate that “if you want to make a tray of ice cubes, start with boiling water” or “the hotter the liquid, the faster it will freeze”. The Mpemba Effect is sometimes presented as a law of nature and not as the curiosity it is.
My proposed experiment would, as skullsinthestars wrote, “take away anything that could cause a Mpemba effect” to show that water under normal circumstances freezes at rates proportional to the sample’s mass, temperature, and inversely proportional to the rate energy is being removed from the sample.
As the article, which I really enjoyed, states: “Later experiments on the effect have been far less conclusive than Mpemba and Osborne’s. Some have seen similar results, while others have observed no effect at all!” I agree with the “number of physicists [who] seem skeptical that the effect truly exists at all”, if the Mpemba Effect is being sold as a fundemental property of water. But if the Mpemba Effect is used to describe two liquids freezing at different rates because their composition, their mass, one is agitated while one is at rest, or that they they are in different freezers of different temperatures, then I agree — of course a sample of pure water will freeze faster than a saturated salt water solution in a -1° C environment, for example.
I didn’t bother doing the experiment because 1.) I don’t have access to the equipment I’d need to control everything to my own satisfation, and 2.) my wife has unrestricted access to Costco, so my freezer is full, too.
I didn’t mean to trash your article, skullsinthestars, or the researchers, or the esteemed journals. But journals have been wrong, even The Lancet. (see http://www.thelancet.com/journals/lancet/article/PIIS0140673697110960/fulltext). I did get the point of the theoretical explanations and agree that there is something at work here other than water and temperature.
My comment was not meant to be taken as the last word on the subject… it was a comment on a blog. As the title asks, “Can hot water freeze before cold?” Sure it can, but not as a general rule. And in spite of 4 citations in 3 different journals, the scientific efforts E.B. Mpemba, D.G. Osborne, and other professional scientists (and perhaps even Baskin and Robbins), the effect is neither reliably replicable nor explained.
Fair enough. It is certainly more subtle than most folks would consider it to be.
Fascinating. I have never thought of it. It makes very little sense… Makes me think though.
Thanks for sharing.
You’re welcome! 🙂
Pingback: Mpemba’s baffling discovery: can hot water freeze before cold? (1969) | Travel to Kelantan
The technician’s attitude towards the result of the experiment may have been developed so because he must have been preached to shoot facts to suit theories instead of theories to suit facts. Its a wrong approach towards things and eventually becomes a habit to ridicule questions and suggestions. Many young minds are tamed because of the attitude. We should be reasonable with them for putting an unusual query. Though unable to answer but to say a straight No is just discouraging.
Its an interesting effect – Mpemba’s Effect. I am surely going to try this one because even I have made ice-creams at home but never came across this phenomenon.
Agreed. The problem is not the technician. The problem is the environment in which the technician was gradually brainwashed by their superiors to keep going until they found the desired answer. The system is broken, not the one person.
In fairness, I would imagine the technician simply thought that he had made a systematic experimental error; that is, that the experimental setup was incorrect and giving an inaccurate result. This is quite common in physics experiments and one has to work hard to make sure that a poor experiment design isn’t leading to a wrong result.
I try not to think of folks as being “brainwashed”! In a sense, resistance to new ideas is a part of the scientific method, which is rather conservative. If an idea comes along that seems well outside of accepted scientific knowledge, scientists will naturally demand very definitive evidence to accept the changes. This can backfire at times, but also protects scientific progress from being led completely astray by any individual anomalous result (such as those caused by systematic error).
This effect, however it is resolved or future developments, shows what amateurs (which includes students not yet blessed with suitable degrees) can accomplish in science. My congrats to Mr. Mpemba for noticing this anomaly at a tender age, and for persisting since amateurs are often ignored. For a potential other example involving disproof of the popular quantum decoherence interpretation of the measurement problem, see name link.
Interesting article. 10 points 🙂
Thank you much!
Thank you for this brilliant post. As a former educator I love the notion of a Hippocratic oath for teachers. It’s imperative not to ridicule–essential to encourage questioning–testing–exploring!
And congrats on being freshly pressed!
Kathy
Thank you! I do believe it’s important to try and encourage questioning, especially in science classes. This is probably one of the most important lessons to learn from science, really!
It is only when there are patient, farsighted and motivating teachers like Osborne that the spark of creativity and genius in children is not suppressed. Also Mpemba stood cool and resolute amid the ridicule that he faced. Nice, inspiring post:)
Thanks! Yes, the determination of the student and the patience of the teacher are both great, inspirational examples.
Pingback: Lipsticktree
It struck me that perhaps atmospheric pressure, height above sea level or below it, might have something to do with the experiment. Remember, for instance, tea does not taste like it should in passenger aircraft, because the water does not reach boiling point except at low flight levels. Could this, by chance, ……? What were the Tanzania test conditions ?
I wondered about similar things myself! Taking a quick look, it seems that the country has a range of ASL height, from 5900 m above sea level for Mount Kilamanjaro and 0 m at the ocean. Another factor that comes to mind is simply the water quality: I can imagine that water in Tanzania in the 1960s was not particularly pure, and it is always possible that the nature of the solutes had a big effect.
🙂
I’ll take that as a “like”!
This read is fascinating and so readable. Are you a teacher? I cannot help but think of my father who made his career in cooling systems for NASA. I would love to have shared your article with him. Thank you for your article that return my thoughts to him thereby giving me a glimpse of things he probably thought about.
Thank you! I am in fact a professor of physics; I like to use my blog to practice making complicated scientific concepts understandable (and light on math), since it is very easy to lose sight of the reality that not everybody enjoys calculus!
I’m glad you enjoyed the article, and I’m glad it helped give you memories of your father.
I remember Bernard Levin, a journalist writing with The Times newspaper, remarking in a letter to The Times at around this time that since he had discovered this effect in noting how hot water freezes faster than cold when thrown onto a frosty road surface, jokingly suggested that this should be called the ‘Mlevin effect’. I think Mpemba is a much more memorable name!
Ha! It is interesting that you mention letters to the paper. Apparently soon after the Mpemba/Osborne paper was published, many people wrote letters to the journals stating that they had either observed the effect themselves in a non-science setting or that it was common knowledge in certain places in the world. A 2006 paper by Jeng on the Mpemba effect summarizes these observations, and a Dr. Kell, who also independently confirmed an effect, noted that in Canada, “Some will say that a car should not be washed with hot water because the water will freeze on it more quickly than cold water will, or that a skating rink should be flooded with hot water because it will freeze more quickly.” Actually, considering I figure skate as a hobby, I now have to ask the rink managers whether they use hot or cold water to freeze the ice!
M. Jeng, “The Mpemba effect: When can hot water freeze faster than cold?” Am. J. Phys. 74 (2006), 514-522.
G.S. Kell, “The freezing of hot and cold water,” Am. J. Phys. 37 (1969), 564-565.
Thank you for telling this story. It is wonderful, and I will be sure to use it with students in the future. What a great way to inspire young scientists.
You’re very welcome, and thank you!
I am amazed at the Mpemba effect, that of boiled water or boiled milk & sugar actually freezing before cold water or cold milk & sugar, is still being ridiculed. Hey, if it works even half the time then it is true! People can be such Absolutists. The story of Mr. Mpemba’s observations & discovery should be known to all as an example of ‘Thinking for Yourself’ rather than becoming a Parrot of somebody elses’s assumed wisdom or values. Too often, Science has become a Religion not to be tested & re-evaluated. Just look at Big Medecine’s acceptance of the Nuclear Power Industry whose reactors spew Cancer causing Radiation continually! We need the Freedom to challenge ideas & debate in all areas of society. I say “Question Everything.”
I don’t know whether I’d say question everything, but there is an excellent lesson that nature does not always conform to our intuitive, and even simple physical, models!
It is too bad people don’t apply Mpemba’s critical thinking skills to all the hoax emails that they forward to me.
Has the Mpemba effect been featured on Mythbusters?
I don’t think it has been done on Mythbusters, to my knowledge; I was thinking that someone should send it along to them!
I want to go over the basic logical case for surprise at this Mpemba effect, as a basis (to any extent needed) for working on understanding it: once the originally hot fluid F2 cools to the same temperature as the comparison fluid F1 that was already cooler to start with, it intuitively seems that F2 should be just like (?) F1 when we first started cooling F1. Let’s say it takes a time delta t sub 1 for F1 to freeze. Let’s say it takes a duration delta t sub 2 for F2 to cool to the original temperature of F1. Then under simplistic assumptions it “should” take a total time delta t sub 1 + delta t sub 2 for F2 to freeze, longer of course than for F1. If that doesn’t happen, then something has to alter the character of F2 so it isn’t just like F1 at the same temperatures. The effect of starting cooling of the hot liquid has to be a continuing alteration, which is interesting and challenging.
In any case, the ME shows that we should be alert for actual empirical findings, and not let theoretical presumptions bind our thinking and observing.
That is probably the central observation here! The effect is only truly surprising if one assumes that the heated water has no other change to it other than temperature. This is why researchers have focused their attention on the properties of solutes in the water.
Wow!!! All I can say is when I was in High School (the late 60s) a science teacher presented the idea that the speed something froze had to do with the original temperature. I can’t remember the entire theory, but after class I asked the question. Based on his theory, wouldn’t boiling water freeze faster than cold water as the molecules would be sped up and so would get to freezing faster (or something like that). My teacher was NOT conducive to questions, so I was dismissed. It stayed in the back of my mind for decades, so I was very excited to read this blog post. I didn’t understand much (not your fault, mine), but net/net….I was RIGHT!! Now, if only I could remember my teacher’s name!!
It’s a little more complicated than that, as your teacher was more or less stating Newton’s law of cooling: that the rate of temperature change is proportional to the difference in temperature. Newton’s law would imply that both samples would cool at the same rate at 30 degrees, and that necessarily the hot liquid would freeze later. However, it sounds like your general intuition, that there might be something different about the hot sample, is on the right track!
This is a brilliantly told story! It was also covered back in the 70’s or 80’s by Jearl Walker in his amazing book, The Flying Circus of Physics. Check it out for many other interesting and mysterious everyday phenomena.
Thanks! I’ll have to look it up.
This effect have been thinking about this effect for sometime. Why are they measuring water using its volume as opposed to its mass? The volume of a quantity of water depends on its temperature. Furthermore how is it that they determine that the water is frozen?
I think in the original Mpemba/Osborne experiment, they started with two equal volumes of water with equal temperature, and then heated up one sample — you are absolutely right that the volume depends on temperature (I was boiling water for a hummingbird feeder the other day, and was struck by how much the volume changed!).
In the early experiments, it seems that freezing was characterized by the first significant accumulation of ice in the sample. In more modern experiments, things have been done more precisely by measuring the temperature of the sample as a function of time. The observations more or less clearly show that the hot samples can beat or meet the cold samples on the race to zero.
With more detailed measurements that show the effect of supercooling, one can actually see the moment of freezing reflected in the temperature plots! In supercooling, the water temperature drops below zero but spontaneously jumps back to zero degrees when rapid freezing initiates. This provides a very clear marker of the moment of freezing!
Nice blog and what a post. I’m gonna follow this post. The article on warm and cold water freezing made me think a lot.
Thanks!
Does this in any way have anything to do with why the hottest deserts can also be some of the coldest places on earth? Or rather, the temperature range that they display…Just sayin…
I think that has more to do with the heat capacity of the air — humid places, with lots of water in the air, can retain more heat and tend to warm up/cool off much slower. I’m certainly not an expert on climate science, though, so I can’t say for certain!
On the flip side of the issue, I know I’ve heard for years that cold water will boil faster than warm water. That said, I’ve never sat down with measurements, themometers and timers to test the theory/hypothesis.
However, as a rule I do try to use very cool water when I want to boil it and I often find myself in a quandry concerning freezing hot items knowing that it freezes quicker but fearing that other items or the freezer might be harmed. I never knew it was a new scientific finding though – or had a name!
Neat!
Someone mentioned something similar to me recently, though I haven’t read anything in the scientific literature about it!
I have seen the “Mpemba Effect” used to argue that there are things that we don’t know about the universe, about science, or about physics in particular … more specifically to make the argument that basic physics (which argues that there is no Mpemba effect) is fundamentally wrong and therefore can’t be trusted. This is then used as the basis to question C14 dating, parent-daughter dating and the age of the earth, etc. etc. and ultimately to support “Young Earth” creationist arguments. Putting it a slightly different way, one person’s great example of pedagogy, which you do here in a wonderful and highly instructive way, is another person’s pedagogic nightmare (I’ll wager this post gets included in home schooling creationist resource lists before long!).
If any creationists try and cite me as an example to support their nonsense, they’ll find me a rather nastily unwilling participant! 🙂 I’ll probably write a follow-up post addressing that issue and link back here for future reference. If you have any links to the creationist “claims” on the issue, I’d appreciate it if you’d send me an email.
It is a gross misinterpretation on their part to argue that the Mpemba effect violates “basic physics”. The “basic physics” in question is Newton’s “law” of cooling, that Mpemba himself refers to, and it states the following: “the rate of change of the temperature of an object is proportional to the difference between its own temperature and the ambient temperature”. The constant of proportionality depends on the material. This would seem to be violated by the Mpemba effect, which considers two “equal” containers of water at different temperatures. However, assuming the “solute” argument holds, the two bodies of water are different, because one has had the material in solution removed. In this sense, there isn’t even a contradiction with Newton’s “law” of cooling, because we’re talking about two subtly different materials.
Not that this matters, either: Newton’s laws were postulated, well, in Newton’s time, c. 1700! Physicists are well aware of the fact that heating and cooling can be a much more complicated phenomenon and not follow Newton’s “law” at all! (Hence I put “law” in quotation marks.) The existence of convection currents, as also referred to in the post, is a situation when the cooling behavior can be dramatically different.
It’s funny that creationists use the very obscure Mpemba effect in their arguments; there are plenty of far more important things not completely understood in physics, such as the foundations of quantum mechanics. My guess is that it seems like such a mundane and ordinary phenomenon that they can say, “See? Science cannot even explain this simple thing!” Hopefully this post shows, however, that we actually have some very good ideas of how the Mpemba effect can occur — it is hardly as mysterious as the creationists would like people to think. If anything, we’ve got too many possible explanations, and it’s a challenge to isolate what combination of these works!
Perhaps they also like to use this effect as an example, though, because of its obscurity. Relatively few physicists are aware of it — quite frankly, it isn’t very important to our understanding of nature! If more scientists are aware of what the Mpemba effect is all about, it will hopefully help refute the nonsense.
Sure, the Mpemba effect being unexpected doesn’t support any particular unrelated claim, or cast doubt on beliefs and general pictures that scientists have triangulated from many approaches. However, it does caution us that “to assume makes an a-s-s out of u and m-e.” Those declarations from some that Erasto had to sadly struggle with, that his results had to be wrong and they’d keep trying until they got the “right” result, etc, are outrageous.
I promise that if I ever see this specific post cited, I’ll let you know right away!
As far as obscurity goes, it is not obscure a all. Everyone “knows” that hot water freezes before cold water. The Mpemba effect is merely a version of that which is linked to a very interesting story. The fact that there are peer reviewed sources to cite is the reason that it is specifically cited in creationist science projects. Also, it seems to address thermodynamics which is at the heart of the “physics” of young earth creationism.
I’m not sure that *everyone* knows, as the popularity of this post seems to indicate that a huge number of people were quite surprised by the result! I can see that the connection with thermodynamics would be appealing to desperate creationists.
I was thinking the same thing. When my creationist brother gets a hold of this, I am never going to hear the end of it. However, the real lesson here is the intellectual honesty of going back to the evidence, which trumps intuition any day. That will be my answer to my brother (who thinks special creation is more intuitive than evolution). By the way, Greg, your blog is great, and I had to read all of ‘Congo Memoirs’ in one sitting because I could not pull myself away.
I hope you can also use this article to refute your brother! As I’ve hopefully made clear, it was recognized right away that this effect didn’t represent dramatically new physics as much as it represents a refinement of our understanding of existing physics.
Of course, creationists have a particular mindset in which anything can be used to validate their views. It probably wouldn’t matter what I said about Mpemba, and they would consider it a “win”. I refuse to tailor my scientific discussions for fear of a creationist distorting them — I wouldn’t be able to talk about anything!
An absolutely fascinating post!
Consider me a subscriber
Thank you very much!
I remember my mom saying something similar in the 70s whenever I was making ice cubes. My dad – and later my husband -scoffed at such an idea. Apparently, she was right – most of the time.
I may be planing memories in my own head, but the more I think about it the more I recall it being a matter of folk wisdom when I was growing up. It seems that a lot of people have had similar experiences!
I see here and other claims, “everybody knows this” or lots of us, but I didn’t. I have some memory of hearing of similar, but it just wasn’t in my awareness per se. After all, consider the power of the counterargument as I outline above: it really is a weird outcome in simplistic terms, and how does the originally hotter liquid “remember” to continue to cool faster toward freezing?
I pointed out in another comment — it apparently isn’t known by everybody, gauging by the response to this post!
Your dad was later your husband?
Pingback: why’o’why | NoMad – zapis wędrówki
Your list of hypotheses is all that is needed to design an experiment to test the contribution of each one to the end result. The experiment would be simple and can be performed at any laboratory.
I like the convention hypothesis. Not only it maintains hot water on top, but it would also cause the liquid to cool more homogeneously oppose to a gradient of temperature. My hypothesis would be that heat transfer is faster when temperature differences are greater. The colder water lacking a heat convention would develop a temperature gradient, colder on the container walls to hotter on the center. This would effectively present colder water to the freezer causing a slower rate of heat transfer than the one presented with convention. The hotter water, with its heat convention, would present a greater delta in temperature between the freezer and the liquid for a longer period of time, and therefore having an higher average heat transfer rate, and thus freezing first.
I know your main topic is how we approach science education and student’s discoveries, but the topic chosen is extremely interesting.
It turns out to be a surprisingly finicky experiment, in part because nobody knows even what starting temperatures would optimize the result. To give an extreme example: clearly, if we start one liquid at 99.9 C and the other at .1 C, the .1 C liquid will almost certainly freeze. At another extreme, if we start one liquid at 31 C and the other at 30 C, we’ll also almost certainly see the 30 C freeze first in accordance with the law of cooling. Especially with convection involved, the size and shape of the containers may play a significant role as well. That isn’t to say that it can’t be done, but those that have tried have run into more difficulties than expected!
I see what you’re saying. There is no “simple”. I meant that it can be done with the equipment found in most labs. However, there are a lot of variables to control.
As I mention, I think it would come down to average rate of heat transfer. Given an amount of heat to dissipate and given a the average heat transfer rate, you can “predict” the time each liquid will take to freeze, everything else as a controlled variable.
I would be nice to try and design such an experiment.
” I meant that it can be done with the equipment found in most labs. However, there are a lot of variables to control.”
You’re right — there’s no particularly complicated equipment needed! The real trick is, as you note, figuring out what parameters need to be controlled, and how.
My father told me this as a wee little lass. He is smart. 😀
Obviously, and he clearly passed it on to his daughter! 🙂
What an excellent article – joy to read. I’d vaguely heard of it before, but never properly read up about it. The story is really one of understanding of the benefits and limits of the scientific method, as much as it about the Mpemba effect per se.
A worthy Freshly Pressed. Nice one!
Thank you!
I seem to remember a suggestion that there is an entropic contribution to the effect, due to the fact that water is densest at about 4 C. As a result, the orientations of the hotter water molecules are closer to the configurations that water needs to get into to form ice. It may thus be that as the liquids are cooled, the initially colder liquid samples molecule configurations more slowly, being trapped in a local minimum in the free energy landscape, whereas the hot water is more likely to end up in a metastable state in which the molecule configurations are “closer” to the configurations need to form ice. Hence the hotter water gets a jump start on ice nucleation.
Of course, other effects could easily change the free energy landscape such that this effect, if it occurs at all, may not be significant.
There is a discussion in one of the papers about various thermodynamic variables — I specifically remember the enthalpy being discussed. It didn’t seem like a deciding factor, so I didn’t discuss it in the post — especially since I figured it would be hard to explain!
Pingback: Top Posts — WordPress.com
As general puzzlement: I see some reference to “water” as exhibiting the ME, but it seems to me more understandable if complex suspensions like milk show it since the purer the water, the harder it is to imagine why it would not just “start over again” as if the cold water, after the delay of reaching that lower temperature (which as I explained, has to give a longer total time to freeze if the substance doesn’t maintain some trace of being affected by previous events. How could rather pure water do that? Shades of “water memory” etc – unless the surrounding environment is the key instead.)
BTW, hat tip to skullsinthestars for being so involved in his (?) thread. I couldn’t be that hard working, it’s great.
I agree that it makes more sense the less pure the water is, and it would seem reasonable that milk, like Mpemba originally used, would be best. Of course, as you’ve noted, it could be more environment/current/temperature distribution related, and the solutes could be a red herring.
And thanks for the hat tip! 🙂
Pingback: Mpemba’s baffling discovery: can hot water freeze before cold? « John Jacob H's RKBA Commentary
Pingback: Links – June 2, 2011 | C6-H12-O6
Looks like Greg Laden (comment posted above, 12:58 June 2) was right that your post is going to be used in unexpected ways. The pingback about two posts above this one goes to a right-to-bear arms website…
By the way, I nevertheless agree with you completely that bloggers shouldn’t self-edit their posts to keep from giving creationists (or other loonies) ammunition. Honest recognition of the limitations of science is the hallmark of a good scientist and the only way knowledge can advance. We question everything, and that is good. The contrast with creationists is stark. What creationist ever points out the errors in the bible?
Well put! 🙂
It helps me to consider the limiting case of two plates filled with a thin layer of water, one nearly boiling and the other cool, both placed in a freezer ( wooden plates, say). It seems clear that evaporation will occur more rapidly from the hot plate, making the hot water layer even so that it could easily freeze before the thicker layer of cool water…not to diminish the many other factors that may play a role, but this makes it clear to me that the Mpemba effect is real …full disclosure, I’m on the record touting Mpemba as one of my physics heroes,.. http://Www.spsnational.org/radiations/2006/director_f06.PDF
Thanks for the comment! That’s a nice thought experiment to demonstrate the plausibility of Mpemba-type freezing.
And I would agree: IMHO Mpemba is a great model for what a physics hero should be!
hmmmmm.. wud love to be able to test this with time lapse photos with IR sensors… or better yet video tape it in IR =D
It would be awesome to see this in a time-lapse image! If I could ever get enough space in my freezer, I might be tempted to get an IR camera and test it!
Hot water cannot freeze. Any frozen water is really cold. I am quite sure about this.
That much is clear! 🙂
With an astronomical amount of pressure, even hot water can be force to solidify =)
Pingback: ResearchBlogging.org News » Blog Archive » Editor’s Selections: Physics of The Bends, Modelling Extinctions from Habitat Loss, and Mpemba’s Effect
Pingback: General updates 2011: May/Jun « The Outer Hoard
Pingback: Lets Talk Geek Episode 51: Antimatter captured iMaverick | Let's Talk Geek
Pingback: The Giant’s Shoulders #36: The ABCs of the History of Science « The Dispersal of Darwin
What a lot of answers. Since it is such an easy experiment to do, I think I will try it but I will put the hot in first, and time it to freeze, and then use the same container and do it at different temperatures.
This reminds me of my friend who claimed he could change ice to boiling water merely by the power of his concentration. After some questioning he admitted that he had only partial success and was only able to raise it to room temperature!
Thanks for commenting! Please feel free to post the results of your experiments here, when you’re done!
Ha!
You could probably make ice to boiling by puting it in vacuum. So there’s some truth to that. If you “concentrate” the pressure else where.
I’ve always been wondering if there were a greater abundance of D2 atoms in the hotter water. Especially since pure deuterium can freeze at 3 C and also takes more heat to boil deuterium. So when you boil the water you lose only H20 to evaporation but would never lose any D2. Or maybe the heat itself can possibly create more D2O? And I also know deuterium has a higher pH so you could also check for that too maybe. But I wouldn’t know. I’ve never gone to school and I don’t have a pH meter or any inteligence to conduct an experiment. But I know I would also like to test 2 bodies of hot water, one in a closed system and one in an open system and see which one freezes faster. I also know that if you throw the hot water out of the cup into the air it freezes instantly. Probably because it thins out, but it still freezes faster than the cooler.
Hmm… interesting thought! I don’t know enough about the properties of D2O to speculate for certain, but it is a unique take on the problem. (It also fits with the general observation that the state of a thermodynamic system is often characterized by more than just temperature.)
Awesome. Well let me know if you find anything. And mean while I’m going to freeze hot and cool water on 2 glass plates and try and study the crystalizion. I noticed last week that hot water crystalizes differently than cool. When I rinsed out two beer glasses one with scolding water and the other with normal temp of the cold side, and there was a noticable difference in crystalization. I just wish I had fancy microscopes that could take pictures. So any opinion or advice on scientific method would be greatly appreciated.
I think for the hell of it I’m going to check the conductivity of hot water gone frozen against cool water gone frozen. And I think I’m even going to place magnets under each glass plate and see if that affects crystalization. The reason why we don’t know this phenomenon is cause we’re not testing every aspect. I want to build a picture from the proton to the electron to the molecule in every way possible. From the entropy to the calculated enthalpy, there will be a formula for this.
Unrelated to water. Actually it in a way is but anyways, I’ve always wondered if particle spin could declare and specify, that particles dimension? The way it spins whether its integer or not. 1/3 or not. Think about it. Particle spin = dimensional energy. Or would it be particle energy equals dimension?
Holy shit I can’t believe I wrote the same exact thing as that gary white guy. Wow! Next time I think I will read everybodys posts.
Pingback: TEDxDar – Who Killed Zinjanthropus?
Pingback: Best Science Writing Online « OPTICS & MODERN PHYSICS
Pingback: Can Hot Water Freeze Before Cold Water? The Mpemba Effect | QED Insight
When I was first faced with this proposition, my first thought was the natural one: the boiling water had to pass through a state identical to that of the cool water before it could freeze.
And that seems to be taken as a given in most discussions.
But it’s not true, unless both waters are being constantly agitated while freezing, because the initial states of both waters are that they have constant internal temperature, but from that point on, they have an internal temperature gradient, and speaking of the “temperature” of the waters is meaningless, since it depends where you stick the thermometer.
So my second thought was to hypothesize that sharper temperature gradients were somehow better for crystal formation: perhaps water molecules moving over an ice crystal are more likely to find a suitable point to which to adhere, than mostly-stationary molecules at the same temperature.
This hypothesis is disprovable, if the effect were no more more noticeable in narrow containers than wide ones; or in thermally conductive containers rather than insulating ones; or if the effect were still observed when the liquids were constantly agitated while freezing.
Sounds reasonable! The fascinating thing about the effect is that there are many possible factors influencing it, and more than one may play a significant role.
You have said that the volume of hot water changes as it cools because the water molecules condense. So, say you take two beakers of water(same amount in each one), boil one and don’t touch the other. The beaker with boiling water should appear to have more in it then the cool beaker. But they should still have the same mass, correct? If you put them in the freezer at the desired temperature, one will freeze faster than the other. Which one freezes first depends on whether the ME worked in your favor, but regardless- when both beakers are frozen they should have the same volume and mass. So in a situation where the ME exists, is the volume and mass the same? Just thinking out loud…
The very last reply I got in my email isn’t here… odd, anyway, it concluded:
“…when both beakers are frozen they should have the same volume and mass. So in a situation where the ME exists, is the volume and mass the same? Just thinking out loud…”
After both are frozen, my personal crackpot theory is that the mass and volume of the ice are NOT the same – the boiling water freezes first BECAUSE it goes into the freezer at a very high temperature and a significant volume evaporates. So much so that what’s left chills down and freezes faster than a cooler container. It should be easy enough to see the net effect – put the boiling and cooler water in containers, weigh, pop ’em into the freezer. You don’t have to see which freezes first, just check them both after they ARE frozen. The problem I have doing the experiment is that someone has a bunch of food in my experimental freezer, its hard to find two locations somewhat removed from each other but clearly similar enough to run the test. Especially getting the same material, volume, etc, for what the sames rest on.
The neat setup is probably to get two 2 cup pyrex measuring cups, they taper slightly so the frozen water should just squeeze up in the middle- no broken glass.
You’re saying my hypothesis is disprovable? As far as freezing water. I mean on a tilted slide. Once we gather the crystalization difference from the two(which there is different crystalization formation.) We can then start studying the isotopes and ionization. For all we know, oxygen itself may play the biggest factor in this.(If you change it’s structure due to heat. And apply some stoichiometry to this phenomena.
I just feel that it might be a little more quantitative. More microscopic than macroscopic. Can you atleast rule that out? I’m not a scientist, I do not have lab equipment nor knowledge. I just started teaching myself science less than 2 years ago. So can you guys atleast try to think outside the box?
Pingback: Open Lab 2012: And the Finalists Are….
did anyone think about all those were not true? Am I the only one thinking that before being at frozen temperature, water goes through the ‘ambient temperature’ of twenty Celcius? therefore how could it be shorter than a previoulsy amiant temperature? If convection is an explanation, convection must have inertia, that is even at 20 celcius, the liquid must keep the convection, I am not sure the energy stored and friction therefore allow for a faster solidification. To me==> urban legend until mythbusters try it.
Myth buster has done this before i believe.
So, 888mlee, what did MBs come up with?
warmer one did freeze i believe
Unexpected “phenomena” occur all the time when conducting experiments. But when things seem to be counter-intuitive you have to examine the way in which the experiment is conducted, the apparatus being used, and what changes are occurring during the set-up of the experiment.
So why is it that SOMETIMES the hotter of two identical water based liquid turn to ice more quickly when placed in a standard refrigerator freezer? Answer: when the following conditions are present; 1. The containers are open – a significant amount of evaporation will occur in the hot liquid – you will end up with less mass to freeze, AND/OR 2: when placing any hot object in a refrigerator you will more likely thaw out any frozen water crystals forming an insulating layer between the metal in the refrigerator and the container containing the liquid thus creating an even greater DELTA-T and smaller R value between the freezing surface and the container. Readers do the same experiment using a closed container starting with the same liquid place one in hot water for any amount of time – to raise the temperature. Dry the outside of the container. place the containers on a piece of insulating material (styro-foam) make no contact with the sides of the refrigerator. The hotter liquid will never freeze before the cold one. This is a controlled experiment that takes into account mechanical conditions and evaporation.
Many years ago as a young man I read an account of a building being shelled during the siege of Stalingrad. Water sprayed from a couple of burst cold and hot water pipes and ran along a wall at an outside temperature deep below the freezing point.
The author observed that oddly enough the cold water ran further while the steaming hot water soon stopped to form into thick round clumps of ice. I forgot his name and that of the book but the scene came back to me much later when I tried to wash the drive before my house clean of its snow cover and correct my clumsiness following up with a bucket of hot water, to see it freeze up even faster. Well in line with this early observation of Aristotle’s of the fishermen on the ice in his days, who secured their rods pouring some hot water around them in the fixing holes they made in the ice.
About 8 months ago I suspended a small cup inside a larger glass bowl in the center. More like a shallow plastic container. Filled the larger bowl with boiling water and immediately ran it out to the -17 degree f average garage freezer above the fridge. Thinking that maybe cooler particles above will drop down. Or even at least condensation. Upon frozen, my shallow dish was fully clear of any amount of water.
This map do not know where we just all here to spend the raise. → freezing temperature is wrong, all the liquid mercury, respectively much lower than water, alcohol freezes -130 -39 degrees doeseoeonda nunohneunnal snow easily because the roots of sand to prevent freezing In contrast, the sand does not freeze well and sprinkle the hot sand, cries Quickly pour cold water to cool down too fast and fourteen reasons to distraction Moved slow-moving molecules, the molecules when moving quickly to meet Moved molecules are moving slowly to slowly grind together As the temperature hits the lower speed is slower These principles in the first place we looked in the refrigerator, which is slightly melted in the refrigerator Put ice cream, put out again after 30 seconds it is fresh On the other hand, melted ice cream, put it this low temperature jineunde Is the state of the clot before the hot water is the same as the second Neotgijeon hot water temperature in the freezer for 70 tao and the most Cold water temperature of 13 degree water at the same time to refinance if you say Put in freezer for 5 minutes in the hot water going down rapidly to 30 degrees The cold water 8 no way the road not different and therefore movement speed of the molecules This is related to temperature and temperature changes caused by the movement of molecules is
A freezer is a territorial place of freezing temperature.
Anything that is found here not to be in a freezing temperature is considered as a foreign object and that that freezing capacity and capability of this freezer would act accordingly without hesitation and delay to protect and maintain its freezing atmosphere.
Now if you put a quantity of very hot/boiling liquid and an equal quantity of cold liquid at the same time, the freezing temperature would not have an equal reaction to both the hot/boiling and cold watrer.
The freezing temperature would first and immediately sense the presence of the the hot/boiling
water and leave behind the cold water.
The freezing temperature would attack first and immediately this hot/boiling water on the ground
that the heat (water vapour) is sensed as a foreign object and is viewed as a threat and therefore must immediately and without delay be suppressed, to guard and protect the freezing temperature of its entire atmosphere.
The freezing temperature would not attack, but instead ignore the presence of the cold water
due to the lack of threatening heat to the freezing temperature of the atmosphere of the freezer.
The freezing temperature will always attempt to equalise or even surpass the amount of heat
it senses from an object to supress it. This amount of freezing temperature it generates will serve
as a blanket for the heat not to leak any further from its container and do any initial damage
to its freezing atmosphere.
While this attack by this freezing temperature to the hot/boiling water is happening at a fast rate, there is no significant amount of freezing temperature applied to the cold water.
Put simply, the boiling water is the enemy and must be put out.
The cold water is not an enemy so it can be ignored.
Analogy:
In a black tribe, try sending two people, one is white the other one is brown.
Who do you think would get the attention first.
The same of what is inside the freezer.
I think story will be a great lesson 4 teachers.Teachers must listen to student’s ideas.
During a cold winter morning in 1985, our hot water line froze (but did not break), while the cold water line was unfrozen. My hypothesis is that heating the water expells the disolved gases (e.g. oxygen). The absence of these disolved gases allows the hot water to freeze, while their presence impedes freezing in the cold water. I have no facilities to test this hypothesis. Perhaps, your readers do.
Very nice article.. I love how lucid some comments are:p truly intriguing concept. I’m doing my final year Physics project on it:)
Pingback: Book review: The Best Science Writing Online 2012 « Wild Muse
It seems that a spontaneously evolving counter current multiplier forms at the boundary layers of both the hot and cold containers. As the hot liquid heats the surrounding air, the warm air RISES and is replaced by more cold air. At the inner boundary, the hot liquid cools and FALLS and is replaced by more hot liquid in a revolving pattern.
Since counter current multipliers become more efficient as the gradient increases, the higher efficiency of the system at the boundary layer of the container with the greatest temperature gradient between liquid and air could be the mechanism for the observed effect.
Disrupting the counter current multiplier by stirring both containers during the experiment and disrupting the air flow within the freezer with a fan should result in the cold liquid freezing first.
The countercurrent thermal exchange system in this case relies on the energy stored in the liquid to overcome the inertia of the the liquid in the container as well as the inertia of the air in the cooling vessel. Though both the hot and cold liquids would evolve their own countercurrent thermal exchange systems, the system in the container with the lower energy would be more sluggish to evolve and the countercurrents would not be as dynamic. The hot container would form an inside out chimney drawing cold air across the outside of the barrier and the inertia of this swirling mass would continue even as the temperature gradient decreased. The hot liquid would do the same on the inside of the container, both sides forming an efficient laminar flow in opposite directions with the temperature gradient as the engine for mass movement.
A spontaneously forming counter current thermal exchange system evolves at the boundary layer between water and air (the container sides) of both the hot and the cold liquid. The energy of the liquid causes the cold freezer air to RISE around the container like an inside our chimney. The cold air surrounding the container cools the liquid closest to the sides of the container causing it to FALL. The liquid and the air are moving in opposite directions ( counter current). The hot water freezes first because the higher energy in that system overcomes the inertia of the liquid and the air quicker and more robustly than the cold liquid. When you place both containers on a rocker and blow a fan on them to keep the counter current thermal exchange system from evolving, the cold container freezes first every time.
Pingback: Advent Calendar of Science Stories 21: Hot and Cold – Uncertain Principles
Pingback: Sternzeichen XY ungelöst - SLEAZEMAG
Great coverage of the history and possible ways experiments can go wrong.
However… when you get down too it, I would argue there is no experiment showing a real mpemba effect with identical containers and pure water. As you rightly discuss, there are many ways experiments on this can fail – by not having pure water, or by not using identical containers, or having the cooled under identical conditions. I would argue that it is actually fairly simple to explain the effect once you look at J. Brownridge’s extremely careful experiments. He found that the effect is due to minute differences in the sample container walls. Even in containers that appear identical, they can have different nucleation sites, which allow initially hot water to freeze first, because it can freeze at a higher supercooled temperature than cold (-4 degrees instead of -10 degrees, for example). If identical containers could be used (or the same container used, and cooled under identical conditions) there is no reason to believe the hot water would freeze first. I have written a blog post on this:
https://moreisdifferent.wordpress.com/2013/04/20/does-hot-water-freeze-faster-than-cold/
Mpemba’s discovery is indeed a very serious accomplishment in African scientific circles. Over the years Africa has contributed in no way in any field in science. This due to high rate of detoriation of facilities in African universities. I hail Mpemba over that feat.
There is one mystery i once observed when i was ten years old in Benue state,Nigeria. I went to the river with my friends to catch crabs. I sighted a brown crab whose body became soft and the other end was turning into a frog. I used a stick to turn it only to have a better look,but it was a dishelled and shrivelled crab metamophorsing into a frog. CAN A TOAD CHANGED INTO A FROG OR TOAD?By my research,i came to realised that any aquatic medium is first colonised by mosquito larva which after some times tadpoles appear,then crabs, frogs,toads and finally fishes. All of them have the same metamophorsis. I need somebody also to help me just like mr osborne.
My unheard of entanglement project:
http://joanne.web.telrock.net
Pingback: Best 14 does hot water freeze faster than cold water mythbusters – ethanadvisors.com