Catching up on some cloaking research that was published after the bulk of my invisibility book was finished — lots of interesting stuff has been happening!
One thing I stress in my discussions of invisibility is that the science and technology is being explored for more than just hiding things from visible light. If we can guide light waves around a central region and send them on their way, we can do similar things with other types of waves. I’ve written before about serious proposals to use cloaking to protect buildings from earthquake waves, and in 2012 a theoretical paper was published about how it might be possible to design cloaking devices to protect offshore structures from ocean waves.
The principles of cloaking have even been used in the design of cloaks for things that are not even waves. A “thermal cloak” was introduced in 2012, which guides heat around a central hidden region, keeping the central region shielded. Similar cloaks have been introduced for static electric and magnetic fields.
I thought I had seen it all when it comes to cloaking, but I was totally surprised when I came across a paper titled “Chemical Cloaking,” published in 2020 in Physical Review E by Avanzini, Falasco and Esposito! The paper introduces the possibility of hiding an object in a fluid flow by using active chemical reactions!
This paper is especially fun for me because it is not only a theoretical paper, but uses very familiar and straightforward mathematics that I could reproduce exactly for this blog post.
So what is the idea of “chemical cloaking?” Let us imagine that we have a steady-state flow of a chemical Z, moving from left to right, as simulated below.

The colors indicate the concentration of the chemical species Z; there is a high concentration on the left, and a low concentration on the right, what is known as a gradient of the chemical density. This causes there to be a net flow of the chemical to the right, as indicated by the arrows. This is a pure, undisturbed flow.
Now let us imagine that we drop some sort of impermeable circular object into the flow; now the chemical must go around the object, causing both the density and the flow of the chemical to be distorted in a significant region around it, as illustrated below.

In principle, if we measured either the gradient or the flow around the impermeable object, we could detect its presence; you can see how the gradient colors are distorted. The object basically causes a “shadow” of flow behind the object on the right.
I have drawn not only the central obstruction in the above plot but also a second circle. Now let us imagine that we can introduce an active region with chemical reactions in the annulus between the inner and outer circle. This reaction might have a chemical formula of the form,
where “A” and “B” are other species of chemicals involved in the reaction. These chemical reactions, depending on the concentrations of A and B, will either add to the concentration of Z or subtract from it. The authors of the paper note that an appropriate choice of the concentrations of A and B at different locations within the annulus can completely cancel out the distortion due to the object; the solution that the demonstrated is illustrated below.
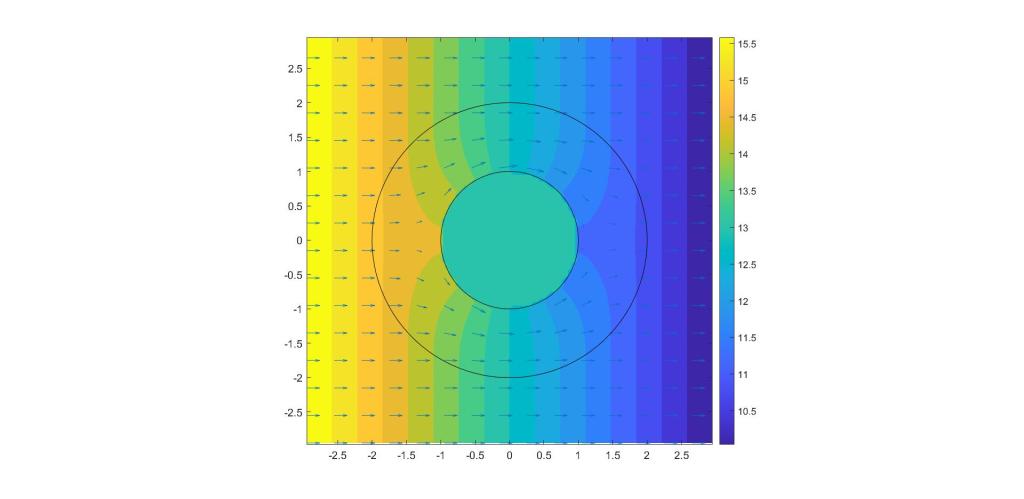
Note that the both the density and the flow of the chemical Z look completely undisturbed outside of the cloaked region! The chemical reactions inside the cloak make the object chemically undetectable.
This is the basic premise of the paper: that it is possible to use active chemical reactions to “hide” a region chemically.
Why might you want to do this? The authors note that cells and cell clusters, for example white blood cells, can sense food supplies or targets by sensing changes in the local chemical gradient. Not said by the authors, but perhaps hinted at, is the possibility of “hiding” therapeutic drugs inside chemical cloaks to prevent them from being attacked by the patient’s immune system. But that it largely speculation: it is not clear how easy it would be to make such a chemical cloak in practice, and considering that chemistry can be very finicky, I imagine that it will be very hard to accomplish.
The authors also note, however, that there might be microscopic organisms like bacteria colonies that have already evolved their own form of chemical cloaking! In that case, this study gives some guidance on what future researchers should look for in the search for natural chemical cloaking.
Overall, this is a really fun and surprising study, and shows that the ideas of invisibility and cloaking can be taken to some very unexpected places!

