This is the second in a series of posts describing the researches of the super-experimentalist Michael Faraday (1791-1867) into electricity and magnetism. In the first post of the series, I discussed his first paper on his electrical research, in which he demonstrated the effect now known as Faraday induction, in which a changing magnetic field can induce electrical currents.
Faraday was, in essence, one of the earliest physicists to actively seek a ‘unified theory‘ of nature, in which all forces of nature are expressed as different manifestations of a single fundamental force. His discovery of induction was a necessary step in the unification of electricity and magnetism, and later work would demonstrate the link between light waves and magnetism (which we’ll come back to in a later post).
The research described in Faraday’s third paper, however, involves unification of a more elementary kind. In this article, Faraday endeavors to eliminate all doubt concerning the idea that different sources of electricity are manifestations of the same fundamental force!
Today, this idea is so fundamental that it is hardly ever mentioned in science classes at all. We know that all electrical phenomenon result from the presence (and motion) of electric charges, typically electrons flowing in an electrical conductor. But the electron would not be identified experimentally until 1896, and in Faraday’s time electrical phenomena fell into three broad categories. “Common” electricity included static electricity, lightning, and electricity brought about by mechanical means. “Voltaic” electricity was that produced by the action of a voltaic battery, and was most associated with chemical action like electrolysis. (Faraday would have a significant impact on electrolysis, as well.) “Animal” electricity was that generated by certain species of animals, such as the “torpedo” (electric ray) and the “gymnotus” (electric knifefish).
In addition to these three, two additional means of generating electricity had been recently discovered: magneto-electricity (Faraday’s induction), and thermo-electricity (electricity generated when a temperature difference is applied across a junction of two metals).
Electrical phenomenon were still poorly understood in Faraday’s time, and these different forms of electricity appeared, at a glance, quite different. For instance, “common” electricity (static electricity) produces a force of attraction/repulsion, while no such force was observed for “animal” electricity. By the time that Faraday did his research, it seems that researchers and philosophers were of the general consensus that the different methods of generating electricity resulted in the same electrical ‘substance’; however, lingering doubts remained. In Faraday’s own words,
The progress of the electrical researches which I have had the honour to present to the Royal Society, brought me to a point at which it was essential for the further prosecution of my inquiries that no doubt should remain of the identity or distinction of electricities excited by different means. It is perfectly true that Cavendish, Wollaston, Colladon and others, have in turn removed some of the greatest objections to the acknowledgement of the identity of common, animal and voltaic electricity, and I believe that philosophers generally consider these electricities as really the same. But on the other hand it is also true, that the accuracy of Wollaston’s experiments has been denied, and that one of them, which really is no proof of chemical decomposition by common electricity, has been that selected by several experimenters as the test of chemical action. It is a fact, too, that many philosophers are still drawing distinctions between the electricities from different sources; or at least doubting whether their identity is proved. Sir Humphry Davy, for instance, in his paper on the Torpedo, thought it probable that animal electricity would be found of a peculiar kind; and referring to that, in association with common electricity, voltaic electricity, and magnetism, has said, “Distinctions might be established in pursuing the various modifications or properties of electricity in these different forms, &c.” Indeed I need only refer to the last volume of the Philosophical Transactions to show that the question is by no means considered as settled.
One suspects, though I am by no means certain of it, that part of the objection to the identification of animal electricity with the other forms had to do in part with resistance to a mechanistic view of living creatures, i.e. the view that living creatures are comprised of the same ‘stuff’ that everything else is made of.
Being the stunningly thorough experimentalist that he is, Faraday decided to compare the various sources of electricity. His experiments focused on “common” and “voltaic” electricity, but he summarized the relevant experiments of others on different electrical sources as well. In Faraday’s day, there were a number of effects which could be associated with the action of electricity, and Faraday classified them as follows:
The various phenomena exhibited by electricity may, for the purposes of comparison, be arranged under two heads; namely, those connected with electricity of tension, and those belonging to electricity in motion. This distinction is taken at present not as philosophical, but merely as convenient. The effect of electricity of tension, at rest, is either attraction or repulsion at sensible distances. The effects of electrical currents may be considered as 1st, Evolution of heat; 2nd, Magnetism; 3rd, Chemical decomposition; 4th, Physiological phenomena; 5th, Spark.
The effects of currents are readily understandable. When electrical current passes through a wire, the wire heats up, i.e. “Evolution of heat.” “Magnetism” refers to Ampere’s law, i.e. the ability of an electrical current to deflect a magnetic needle. “Chemical decomposition” of course refers to the ability to use electric currents for reactions such as electrolysis. “Physiological phenomena” refers to the ability of current, “when strong, to shock the whole animal system, and when weak to affect the tongue and the eyes.” “Spark” of course refers to the discharge of visible light through an electric spark.
Faraday goes on to describe a number of experiments he personally performed relating to “voltaic” electricity and “common” electricity, and sumamrizes the results of other researchers concerning the other types. We note only one of these, which demonstrates Faraday’s strict attention to detail: the discharge of voltaic electricity through heated air.
It was already well known that “ordinary” electricity could be transmitted across a gap of air more readily if the air was heated; Faraday decided to investigate whether “voltaic” electricity would also show increased transmission through heated air. His apparatus is illustrated below, both with Faraday’s original figure and my reconstruction:
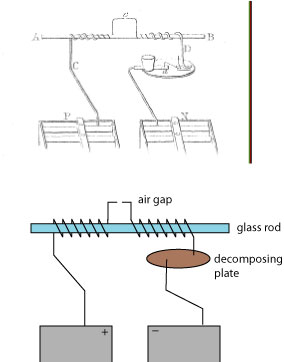
In essence, an electric circuit is formed between the positive and negative poles of a battery, broken only by a small air gap. A “decomposing apparatus” is also inserted into the setup, which is used to electrically excite chemical reactions (described below). Faraday, in his own words, again:
Whilst in the state described, no decomposition took place at the point a, but when the side of a spirit-lamp flame was applied to the two platina extremities at e, so as to make them bright red-hot, decomposition occurred; the heated air was established. On raising the temperature of the points e by a blowpipe, the discharge was rendered still more free, and decomposition took place instantly. On removing the source of heat, the current immediately ceased. On putting the ends of the wires very close by the side of and parallel to each other, but not touching, the effects were perhaps more readily obtained than before. On using a larger voltaic battery, they were also more freely obtained.
Being even more thorough, Faraday replaced his chemical apparatus with a galvanometer as a second confirmation that current had flowed under heating.
It is perhaps worth looking at his decomposing apparatus, as well:
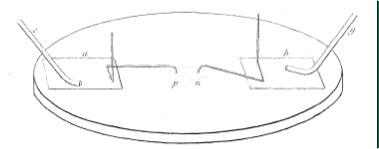
A much more convenient and effectual arrangement for chemical decompositions by common electricity, is the following. Upon a glass plate, fig. 2, placed over, but raised above a piece of white paper, so that shadows may not interfere, put two pieces of tinfoil a, b; connect one of these by an insulated wire c, or wire and string with the machine, and the other g, with the discharging train or the negative conductor; provide two pieces of fine platina wire, bend as in fig. 3, so that the part d, f shall be nearly upright, whilst the whole is resting on the three bearing points p,e,f; place these as in fig. 2; the points p,n then become the decomposing poles. In this way surfaces of contact, as minute as possible, can be obtained at pleasure, and the connexion can be broken or renewed in a moment, and the substances acted upon examined with the utmost facility.
A drop of solution to be decomposed could be placed connecting the p and n poles, thus completing the electrical circuit.
Curiously, he waits to describe the device until much later in the paper, when he studies the ability of “common” electricity to produce chemical reactions. Using a “plate electrical machine”, Faraday charged a series of Leyden jars with “common” electricity, and then discharged it through his decomposing apparatus. His machine “caused the precipitation of so much copper on the end p, that it looked like copper wire; no apparent change took place at n.” A number of other chemical decompositions were also effected with “common” electricity.
One aspect of his electrical device seemed curious to me. To discharge his Leyden jars, he connected the ground wire to what seems to be a rather risky outlet:
A good discharging train was arranged by connecting metallically a sufficiently thick wire with, first, the metallic gas pipes of the house, then with the metallic gas pipes belonging to the public gas works of London; and finally, with the metallic water pipes of London.
I guess it worked out okay for Faraday, but I wouldn’t think to discharge a collection of static electricity anywhere near a source of flammable gas!
Faraday summarized the state of the art in analyzing different sources of electricity with the following table:
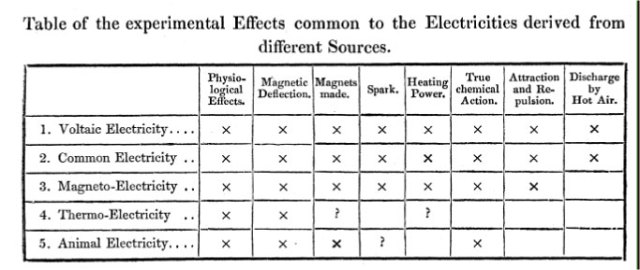
As noted, many of these results were undertaken by other researchers. Also, there are many blank spots on the table. Nevertheless, Faraday felt comfortable in declaring,
The general conclusion which must, I think, be drawn from this collection of facts is, that electricity, whatever may be its source, is identical in its nature. The phenomena in the five kinds or species quoted, differ not in their character, but only in degree; and in that respect vary in proportion to the variable circumstances of quantity and intensity which can at pleasure be made to change in almost any one of the kinds of electricity, as much as it does between one kind and another.
Faraday’s quotation highlights another curious problem of scientific research in his era that is now rarely considered: no fundamental measure of electricity yet existed. Indeed, as we’ve just pointed out, there was still not agreement on whether there was a ‘fundamental’ type of electricity, much less any agreement on how to quantify it.
Faraday clearly understood that the difference in electrical sources was due to the different amounts of electricity in the various sources. But without a standard unit of measuring electrical behavior, one could not easily compare them. Nowhere in the paper is there a discussion of the unit of electric potential (the Volt) or the unit of current (the Ampere), though the work of Ampere is mentioned and the voltaic battery is named after its discoverer, Volta.
To rectify this problem, Faraday made some significant attempts to compare the amount of electricity produced by a voltaic battery and a “common” battery, and these efforts were described in the second half of his paper. Faraday compared the strengths of a voltaic and common battery by their effect on a galvanometer and chemical reactions. His conclusions:
Hence it results that both in magnetic deflection and in chemical force, the current of electricity of the standard voltaic battery for eight beats of the watch was equal to that of the machine evolved by thirty revolutions.
It also follows that for this case of electro-chemical decomposition, and it is probable for all cases, that the chemical power, like the magnetic force, is in direct proportion to the absolute quantity of electricity which passes.
This was evidently one of the first attempts to come to grips with a universal measurement of electricity!
This demonstration of the identical nature of various manifestations of electricity was not the first and would not be the last of Faraday’s unification efforts. We will see some other fascinating results related to what might be called “Faraday’s unified theory” in future posts.
**************
M. Faraday, “Experimental researches in electricity. Third series,” Phil. Trans. Roy. Soc. Lond. 123 (1833), 23-54.


Thank you Faraday for clearing the question on the nature of electricity! I guess we also have to thank him in part for the very neat “wireless” charging dock of the new Palm Pre based on magnetic induction.
It’s surprising that his primary method of detecting electrical current flow was visually observing chemical decomposition! As an electrical engineer, I can hardly imagine how annoying these experiments must have been to perform without a digital multimeter 🙂
It’s too bad Faraday doesn’t get more credit among laypeople for this unification of electricity and magnetism. It seems like Maxwell gets the lion’s share, though he really just mathematized what was already known (except for his addition of displacement current).
Wade: To clarify a little bit: I think the galvanometer was the standard instrument for measuring current flow, which may not have been completely clear from my post. However, because those early experimenters weren’t certain that all electricity was the same, a number of different ‘standards’ were developed for assessing the presence of current. I find this part of Faraday’s work fascinating because it shows how difficult it is to study a scientific topic when almost none of the fundamentals are understood.
“It’s too bad Faraday doesn’t get more credit among laypeople for this unification of electricity and magnetism.”
Yeah, this is a great example of the detailed history getting simplified to the point where it’s incorrect! As we’ll see in a couple of additional Faraday posts, Faraday probably deserves credit as the greatest proponent of unified field theories. His intuition told him that electricity, magnetism and light were all related long before Maxwell formulated his equations.
Pingback: Chapter 6 | Offshore Welders
Just to say thanks – I was looking for Faraday’s unification of electric sources and you have it all. Well written as well.
Faraday was definitely a man many years ahead of his time. We can only imagine the understanding and other hypothesis he could have developed in modern times with modern technology.
Faraday wouldn’t get a job today in a decent university. There’s no place nowdays in Physics for factual and logical thinking, only mysticism and blind-faith are permitted.