So what does an atom look like? If you were to pull someone at random off the street and ask them to draw a picture of an atom, they would more likely than not draw something like this:
Almost everybody knows this picture: negatively charged electrons “orbit” around a positively charged and tiny nucleus under the influence of the force of electricity, very much analogous to the way planets orbit the Sun in our solar system under the influence of gravity.
This image is ingrained in the public’s consciousness — even though it is quite inaccurate! Electrons are not point particles that travel in well-defined orbits around the nucleus; more accurately, electrons act as smeared-out “clouds” that encircle the the nucleus in well-defined distributions dictated by the laws of quantum mechanics.
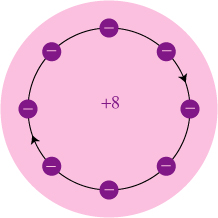
Still, this planetary model is an important one historically, and was accurate enough in its time (and still today) to forgive its faults. It arose naturally in the early 1900s, in a period of great confusion and uncertainty about atomic structure. With tantalizing and rather bewildering experimental hints, scientists speculated wildly about the nature of the atom. The strongest contender was the “plum pudding” model of J.J. Thomson, in which atoms were visualized to be a “pudding” of positively-charged fluid within which were embedded negatively-charged electron “plums”. In Thomson’s original paper, these plums were arranged equidistantly around a circle within the pudding and orbiting within it:
The “plum pudding” model became widely accepted not so much because it answered any nagging questions about atoms, but rather because it conflicted the least with the known physics of the time! In 1909, however, Ernest Rutherford set two of his assistants, Hans Geiger and Ernest Marsden, to probing atomic structure using radiation, in what is now known as the “gold foil experiment“. High velocity alpha radiation was used to bombard a thin film of gold, and the direction of scattered alpha rays was measured. In the plum pudding model, it was expected that all of the alpha rays would be slightly deflected, but that none of them would be strongly deflected, simply because the electric charge is either too spread out (pudding) or too light in mass (the plums) to obstruct their motion. Geiger and Marsden, however, found that some of the alpha particles were actually reflected back towards the source! As Rutherford later commented,
It was almost as incredible as if you fired a fifteen-inch shell at a piece of tissue paper and it came back and hit you.
In a 1911 paper, Rutherford introduced the planetary model of the atom as described above, in which electrons orbit a very small, heavy and dense positively-charged nucleus. The rare alpha particles that bounced backwards were interpreted as having a direct collision with a much heavier nucleus of a gold atom. In 1913, Neils Bohr managed to explain the emission spectra of atoms by assuming that electrons can only circle the nucleus in orbits of quantized (discrete) angular momentum. The Bohr model completely opened the door to the theory of quantum mechanics, which is still used today to describe the behavior of atomic and subatomic particles.
In this standard telling of the story, Ernest Rutherford is given credit as the scientist who “invented” the planetary model of the atom. However, as is true in many stories of discovery, the truth is a bit more complicated! The first mention of a planetary atom in fact goes back to a lecture given in February of 1901 by the French physicist Jean Baptiste Perrin.* Perrin’s lecture** not only illustrates a truly forgotten milestone in the history of physics, but also describes in a clear and non-technical manner the scientific understanding of atoms and molecules at the turn of the century.
If anyone could have guessed at the structure of the atom, it was Jean Baptiste Perrin(1870-1942). Born in Lille, France, and educated at the École Normale Supérieure, he became an assistant in physics at the École in the year 1894. With this position, he began investigating the properties of the mysterious cathode rays (electrons, discovered in 1869) and later Röntgen rays (X-rays, discovered in 1895). Among his early achievements was a demonstration that cathode rays consist of corpuscles (discrete particles) of negative electric chargeHis research was productive; by 1897 he had earned his doctorate and immediately took a position as a lecturer in physical chemistry at the Sorbonne in Paris.
In his new role, he developed an intense interest in the “molecular hypothesis”, the now obvious idea that matter is made up of discrete atoms that form molecules and larger structures. These investigations would lead, in later years, to remarkable and important discoveries and eventually a Nobel prize — but more on that later!
It is perhaps somewhat surprising to realize that, even at the end of the 19th century, the existence of atoms was not yet definitively proven or understood. Since ancient times, naturalists and philosophers had been divided on the nature of matter. In one camp were the atomists, who argued that all matter must be made up of extremely tiny and fundamental particles — atoms — that could not be divided further. In the other camp were believers in the “continuum”; that is, people who believed that matter was infinitely divisible. For several millenia, this debate remained an open an unsolvable one, simply because atoms, if they existed, were too small to be observed with either the naked eye or, later, optical microscopes.
In 1804, things seemed to turn in the atomists’ favor when John Dalton advocated their view with his observed law of multiple proportions:
If two elements form more than one compound between them, then the ratios of the masses of the second element which combine with a fixed mass of the first element will be ratios of small whole numbers.
That is, to use the example from Wikipedia, it is possible to react carbon with oxygen in two ways, one of which forms carbon monoxide and one of which forms carbon dioxide. We can react 100 grams of carbon with 133 grams of oxygen to form CO, and that same mass of carbon can react with 266 grams of oxygen to form CO2. The ratio of masses of oxygen in the two compounds is
CO2 : CO = 2 : 1,
suggesting that oxygen can only join with carbon in discrete amounts.
With a little theoretical imagination, however, one can argue that the continuum theory applies, but that oxygen only reacts with carbon in direct proportions according to some more obscure law. Even in the mid to late 19th century there were proponents of the continuum hypothesis; without direct evidence of the action of atoms, the question remained open.
This is where Perrin’s lecture begins, introducing the molecular hypothesis and noting its limitations. He uses a quite charming analogy to make his point***:
The hypothesis chosen by mutual agreement, as the simplest, is to admit that any pure substance, water for instance, is actually formed by an extremely large number of distinct particles of matter, absolutely identical to each other, which define the extreme limit of divisibility possible for water. These are the molecules of the body.
Let us be clear on what is meant by giving the molecule as the limit of the possible division for the body. A comparison will suffice. Suppose that we perceive in the country a white spot, distant, which can be divided under influences of any kind into spots of similar appearance, but smaller. We will make a molecular hypothesis by assuming that this spot is actually a herd. I do not think it very useful to add thatthis does not mean that a sheep is not divisible into smaller parts, but only that for the division, it will be necessary to go about it other than to split the herd and that the observed phenomena become quite different.
This is a lovely and rather insightful analogy. Perrin points out that (a) just because something appears continuous, like a herd visible from a distance, that does not make it so, and (b) a molecular hypothesis does not imply that the individual molecules or atoms (i.e. sheep) cannot be further divided — such additional division, however, must proceed by very different physics. On this latter point, Perrin wisely leaves the door open for future subdivisions of the atom, exactly what is done in nuclear physics.
But atoms and molecules themselves cannot be directly measured, as Perrin notes:
With this understanding, how can we prove that a pure substance is an agglomeration of discrete particles, identical, and resistant to division, in the sense just made clear?
Direct verification, we have seen, is not currently possible; it remains, according to familiar practice in the Inductive Sciences, to investigate whether, among the consequences of this assumption, are any accessible to experimental verification. Such consequences are numerous, and if we are not able to deduce them from any other hypothesis, we perhaps have no right to say that the molecular hypothesis is true, but at least we know that it is useful.
We can’t directly “see” atoms and molecules, but we can deduce many physical effects that are consequences of their existence. This in itself does not prove their existence, but at least shows that the molecular hypothesis can be a useful tool.
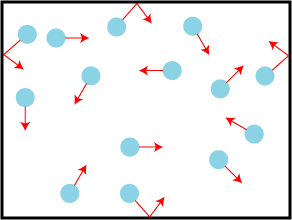
Several examples of such indirect consequences come from the kinetic theory of gases, introduced by Clausius and Maxwell in the mid-1800s. In this theory, it is assumed that a gas consists of a large number of molecules in constant motion at very high speeds. On average, these molecules are assumed to be very far apart from one another, and the volume of the gas is thus much larger than the combined volume of the individual molecules. Molecules can bounce off of each other and off of the walls of the container enclosing the gas; the latter effect is what is perceived as the gas pressure. A final, important, assumption is that the molecules bouncing off of each other and the walls lose no energy in the collisions, which are said to be perfectly elastic.
Perrin explains this idea with an analogy that is still relevant today:
We have a representation, crude, of this hypothesis, by imagining a large pool table on which would rollin all directions a large number of balls. But, and this is why this model is poor, the balls and cushions of the pool table are not perfectly elastic, so that the movement would end soon. However, we can see in this model an interesting result. Among the balls colliding with each other, they also encounter the cushions of the pool table; suppose that we have removed the screws that hold motionless a side of the frame, one of the cushions; this cushion will be pushed back by the balls that they encounter; to keep at rest, it will be necessary to exert some effort: therefore we understand why, for a piston to keep at rest cut in the wall of an enclosure containing a gas, it must exert effort: the effort is needed to counter-balance the impact of the molecules against the piston.
We can immediately derive (as Perrin and others did) one important experimental result based on the kinetic theory. Let us suppose that we have a gas contained to a box of volume V at pressure P. If we stretch the length of the box by 2, the volume doubles to 2V. But the molecules will spread out, and half as many will strike the same area of the walls as before; the pressure will therefore be halved to P/2. We have therefore reasoned our way to Boyle’s law, which states that, for a given temperature, pressure and volume are inversely related:
PV = constant.
Boyle’s law is a special case of the ideal gas law, familiar to high school and undergraduate chemistry students the world over:
PV = NkT,
where N is the number of molecules in the gas, T is the temperature and k is Boltzmann’s constant.
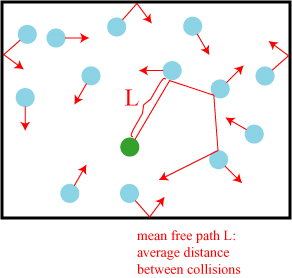
The kinetic theory of gases can lead to an estimate of a surprising number of molecular properties. Though the molecules in a gas are on average relatively distant from one another, they are also moving very fast (“not much slower than a cannonball,” Perrin notes). This means that the molecules will collide with others quite often, and one can determine the average distance a molecule travels before it hits another. This distance is referred to the mean free path, and will be labeled L.
This mean free path can actually be determined using the kinetic theory of gases — a tenth of a millionths of a meter, by Perrin’s reckoning — and once L is known, even more molecular properties can be derived:
Once one knows the mean free path L, it becomes easy to calculate the diameter D of the molecule, and the number N of molecules that are contained at a fixed temperature and pressure, in one liter of gas considered. It can be shown that one can find two equations that determine these two new unknowns.
First of all it is obvious that if the pressure decreases, that is to say if the number N becomes smaller, the mean free path must grow. Likewise, the free path should depend on the diameter D, for, other conditions remaining the same, two molecules that pass each other are all the more likely to collide if they are bigger. By clarifying this somewhat vague line of reasoning, we obtain an equation containing N, D and L, that is to say, since L is already known, an equation containing only the unknowns N and D.
Now suppose, for example by reducing the temperature, it solidifies the gas initially contained in a liter and it then occupies a volume v that can be measured and in which are contained the N primitive molecules. Because of the almost absolute incompressibility of solids, it is natural to assume that these N molecules are bonded against each other in the volume v. The volume v can not differ much from the actual sum of the volumes of N molecules, that is to say of N spheres of diameter D: this makes a second equation between N and D. Unfortunately this second equation is only approximate.
We can therefore, at least in an approximate manner, calculate the diameter of molecules and their number.
In short: bigger molecules must collide more often, and from this one can determine one mathematical equation relating the number of molecules N, their diameters D, and the mean free path L. If we can further freeze our gas into a solid, the volume of the solid must roughly be equal to N times the volume of the individual molecules. With these two equations, we can solve for the number of molecules and their diameters.
In the lecture, Perrin estimates that the sizes D of molecules range between one thousandth of a micron and one ten-thousandth of a micron (where a micron is a millionth of a meter). This is remarkably close to the the sizes we have determined with modern methods!
Furthermore, Perrin estimates the number N of gas molecules within a liter of gas. The number reported in the lecture is 55 billion trillion molecules/liter ( molecules/liter), a stunningly large number and a surprisingly accurate one for the time.
With an understanding of the number and size of molecules, Perrin then turns to the structure of the molecules themselves. According to the ideal gas law, two volumes of gas at the same temperature and pressure contain the same number N of molecules, regardless of the specific molecular properties. But when we combine a liter of hydrogen H and a liter of chlorine Cl to form gaseous hydrochloric acid, an interesting thing happens: we end up with two liters of acid, and therefore twice as many molecules of hydrochloric acid, or 2N molecules of HCl. Perrin correctly deduces that a molecule of gaseous hydrogen in fact consists of two atoms of hydrogen combined, with a similar argument for gaseous chlorine. A molecule of hydrogen and a molecule of chlorine therefore combine to form two molecules of hydrochloric acid. In chemical notation, this would be written as
.
Something more unusual happens when we put mix salt — sodium chloride, NaCl — in water. A mixture that begins with N salt molecules freezes as if it contains 2N molecules; this, however, can only happen if the sodium Na and the chloride Cl are disassociated in the water! This is particularly surprising because it was well known even in Perrin’s time that pure sodium basically blows up when put in water! Evidently sodium disassociated in water via salt behaves very differently than pure sodium just tossed in a bucket of water.
The difference lies in the electrical properties of the dissolved salt. Sodium chloride in water forms positively-charged atoms of sodium and negatively-charged atoms of chlorine; these are what we know as ions. This disassociation into ions can be demonstrated by using electrodes dipped in the solution to electrically separate the two species of atoms. Ordinary sodium is not in ionic form, and thus has a different reaction to water.
This discussion of atomic ions leads naturally to Perrin’s own knowledge of cathode rays, i.e. fast-moving electrons, also referred to as “corpuscles”. Through a variety of different experimental results, it is determined that a hydrogen atom consists of a negative electron of very small mass and a positive hydrogen ion of equal and opposite charge and one thousand times the mass. Furthermore, the electron is extremely small compared to the size of the atom as a whole.
These final observations, at last, bring us to the planetary atom, assembling the diverse (albeit indirect) snippets of knowledge discussed in the lecture:
For the first time, we see a way to penetrate the inner constitution of the atom. We will make, for instance, the following hypothesis, which is consistent with the foregoing facts.
Each atom would consist, first, by one or more masses strongly charged with positive electricity, sort of suns whose positive charge is much higher than that of a corpuscle, and secondly, by a multitude of corpuscles, sort of small negative planets, the whole of masses gravitating under the action of electric forces, and thetotal negative charge exactly equal to the total positive charge, so that the atom is electrically neutral.
The negative planets which belong to two different atoms are identical; if it happens that the suns positive are also identical to each other, the entire material universe is formed by the grouping of only two species of primordial elements, positive electricity and negative electricity.
This is quite an astute observation: the idea of identical positive “suns” anticipates the discovery of the proton, the fundamental positively-charged component of the nucleus.
If a sufficient electric force acts on an atom it can detach one of the minor planets, a corpuscle (formation of cathode rays). But it will be twice as difficult to pull a second corpuscle due to the excess of the total positive charge, unaltered, on the negative charge remaining. It will be three times harder to pull a third corpuscle, and when our capabilities are exhausted, we will have almost nothing ripped from the atom, whose apparent indivisibility is thus explained. To tear a positive sun, it would be quite beyond our present power.
Perrin correctly identifies cathode rays as being electrons ripped from the outer “orbits” of atoms.
The atom thus appears as a whole gigantic, whose inner mechanics would have as basis the fundamental laws of electrical action. The duration of gravitation of the different masses internal to the atom may correspond to different wavelengths of light rays that show the emission spectrum.
Here Perrin has the right idea: the wavelengths of light emitted by atoms are directly related to the behavior of the “orbiting” electrons. However, Perrin would not be able to precisely calculate these wavelengths without the quantum assumption made by Bohr. Nevertheless, he made a remarkable estimate****:
A simple calculation provides an initial indication in this direction. Cathode rays detached from aluminum by the ultra-violet light have, according to Lenard, a speed of about 1000 kilometers per second. Suppose that the corpuscles that form these rays have about the same speed in the atoms from which the light has detached, and the time that we seek is set by one of these particles in describing the speed to travel the circumference of the aluminum atom, that is to say according to the dimensions of this atom, about
cm, we find that the duration of gravitation (the year of this planet) is about
seconds. But the periods of vibration of the aluminum lines are between
and
seconds. This is a remarkable coincidence, to my knowledge not yet reported.
This is indeed a remarkable calculation! It is, strictly speaking, not correct, but there is enough accuracy in the assumptions that Perrin makes that he gets in the right ballpark, so to speak.
If the atom is very heavy, that is to say, probably very large, the particle farthest from the center, the Neptune of the system, will be poorly retained in its course by the electrical attraction of the rest of the atom, with the slightest cause it detaches; the formation of cathode rays could become so easy that the matter appears spontaneously radioactive, such are uranium, thorium, which have precisely the greatest atomic weight surely known, such appear to be new highly radioactive metals recently discovered in France by Mr. and Mrs. Curie (1898) and Debierne(1899).
Here Perrin speculates on the nature of radioactivity. Heavy atoms have many, many electrons orbiting them; the most distant electrons would by their nature be very weakly bound to the atom, and Perrin argues that they could be detached with the slighted disturbance, appearing as radioactivity. This is, of course, incorrect; radioactivity arises in the nucleus of the atom, not in the orbiting electrons; we have discussed this in the context of other speculations on radioactivity previously.
Perrin’s speculations are the first known written description of a “planetary atom”, which eventually formed the basis of atomic physics.
It is not difficult, however, to see why Perrin’s arguments didn’t take off. The planetary atom, as Perrin conceived it, answered no open questions about the behavior of matter. Even worse, it suffered from the same limitation that most early models of the time possessed: accelerating electrons, such as those orbiting a positively-charged “sun”, were expected to rapidly lose energy from electromagnetic radiation. Perrin’s simple planetary atoms would collapse upon themselves in a fraction of a second; obviously, real atoms don’t do this. The solution of the problem would be the introduction of the quantum theory; in the meantime, nobody was willing to believe in a planetary atom until Rutherford’s experimental evidence made it impossible to ignore.
Perrin did not suffer from lack of recognition, however; his work on the molecular hypothesis put him in a perfect position when, in 1905, Albert Einstein came up with a theory to explain Brownian motion.
For literally thousands of years, naturalists had observed that nearly microscopic particles suspended or floating in liquids or gases will “dance about”, jittering in a seemingly random motion. The scientific observation of this Brownian motion is generally credited to the botanist Robert Brown in 1827.
It took the genius of Albert Einstein to properly explain the jittering as the result of the visible particle being jostled by the undetectable rapid motions of atoms and molecules, as described by the kinetic theory of gases. Here at last was direct evidence of the existence of atoms and molecules, putting to rest the arguments between the atomists and the continuum advocates. Einstein’s theory provided quantitative predictions for the motion of a “Brownian particle”, and it was Jean Perrin who confirmed experimentally these predictions.
Jean Perrin’s work led him to win the 1926 Nobel Prize in physics, “for his work on the discontinuous structure of matter, and especially for his discovery of sedimentation equilibrium“.
Even though his accolades came for other accomplishments, Perrin was clearly proud of his planetary atom hypothesis; in his Nobel acceptance speech, he said:
I was, I believe, the first to assume that the atom had a structure reminding to that of the solar system where the “planetary” electrons circulate around a positive “Sun”, the attraction by the centre being counterbalanced by the force of inertia (1901). But I never tried or even saw any means of verifying this conception. Rutherford (who had doubtless arrived at it independently, but who also had the delicacy to refer to the short phrase dropped during a lecture in which I had stated it) understood that the essential difference between his conception and that of J.J. Thomson was that there existed near the positive and quasi-punctual Sun, enormous electrical fields as compared with those which would exist inside or outside a homogeneous positive sphere having the same charge, but embracing the whole atom.
Here we see a wonderful graciousness between two important scientists, namely Rutherford and Perrin. Perrin, who was the first to postulate a planetary atom, properly credited Rutherford for the true and proper discovery of the nuclear atom. Rutherford, in turn, was kind enough to note Perrin’s priority in the idea of the planetary atom.
Both scientists were wise enough to realize that physics, and science in general, is a process that involves many researchers and many ideas over extended periods of time. Though Rutherford deserves the credit for the nucleo-planetary atom, others such as Perrin were being led inexorably towards the same conclusion. Science is, in a sense, about inevitability!
************************
* I’ve noted Perrin’s planetary atom theory in a previous post; however, I only recently found his original paper on the subject and had time to translate it from the French.
** J. Perrin, “Les hypothèses moléculaires”, Revue Scientifique, 4th series, Volume 15 (1901), 449 -461.
*** Quoting only selected parts of the complete 12-page lecture. Translations are my own, using translation software and obvious corrections.
**** My translation software failed a bit here, but the meaning of the passage was clear; I adapted it to a sensible English form.





You have no idea! You said “Electrons are not point particles that travel in well-defined orbits around the nucleus; more accurately, electrons act as smeared-out “clouds” that encircle the the nucleus in well-defined distributions dictated by the laws of quantum mechanics.”
Electrons are point particles. The “smeared out cloud” is just the probability density as the absolute square of their wavefunction, which arises due to quantummechanics. What this probability density does is it allows to calculate the probability for the electrons to be in a certain volume by integration of the wavefunction. So not the electrons are smeared out, our knowledge of where they are has the uncertainty. If electrons were no point particles we could never do all the diffraction experiments. So please, before you write about physics, read wikipedia! I couldn’t even continue reading the rest of the article..
Awesome post. Sorry about the idiot in the first post.
Thanks! 🙂
Very sweet reasoning by Perrin. I love it when one get a chance too look behind the curtains of how people though it out. And yes, they had a certain flair those two, and decorum. A calmer time perhaps? I’m having a fine time reading you presenting the atom, and I’m guessing you’re planning to discuss it in more detail later? Like eh, in forms of probability etc? Anyway, we’re star stuff, ain’t we 🙂 all of us.
Thanks! I’ll be talking about more basic quantum in the near future — I’ve been reading the original papers by Einstein, Planck and others to give me a better feel for their reasoning.
Very cool reading.
I’m looking forward to it.
Because thinking is eternal.
It is a very good article I just think the title is a little preposterous. The atom always existed it was not invented it was DISCOVERED.